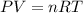Chemistry, 05.03.2020 20:07 nathanscastr02
In preparation for a demonstration, your professor brings a 1.50−L bottle of sulfur dioxide into the lecture hall before class to allow the gas to reach room temperature. If the pressure gauge reads 173 psi and the lecture hall is 20°C, how many moles of sulfur dioxide are in the bottle? In order to solve this problem, you will first need to calculate the pressure of the gas. Hint: The gauge reads zero when 14.7 psi of gas remains.

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 19:00
Asyringe contains 56.05 ml of gas at 315.1 k. what volume will that gas occupy if the temperature is increased to 380.5 k? a) 12.41 b) 46.42 c) 67.68 d) 81.74
Answers: 1

Chemistry, 21.06.2019 22:30
What are the charges of the subatomic particles by choosing the answer from the drop down menu. protons have a (+1,0,or-1). (protons,neutrons,electrons) have a 0 charge. 3.) electrons have a (+1,0,-1)
Answers: 2

Chemistry, 22.06.2019 07:40
21. consider the following chemical reaction: n2+ o2 2 no if 10.0 g of n2 reacts with excess oxygen then how many grams of no can be formed? a) 10.7 g b) 21.4 g c) 32.9 g d) 42.8 g page 4 of 8
Answers: 2

You know the right answer?
In preparation for a demonstration, your professor brings a 1.50−L bottle of sulfur dioxide into the...
Questions

Geography, 21.04.2020 11:09




Mathematics, 21.04.2020 11:09


Mathematics, 21.04.2020 11:10


Chemistry, 21.04.2020 11:10


Mathematics, 21.04.2020 11:10

Mathematics, 21.04.2020 11:10


Mathematics, 21.04.2020 11:10


Chemistry, 21.04.2020 11:10


Mathematics, 21.04.2020 11:10

Mathematics, 21.04.2020 11:10