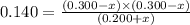Consider mixture C, which will cause the net reaction to proceed in reverse. Concentration (M)initial:change:equilibrium:[XY]0 .200+x0.200+x←net⇌[X]0.300−x0.300−x +[Y]0.300−x0.300−x The change in concentration, x, is positive for the reactants because they are produced and negative for the products because they are consumed.

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 13:10
What type of interaction occurs between the r groups of valine and isoleucine in a tertiary structure? view available hint(s) what type of interaction occurs between the r groups of valine and isoleucine in a tertiary structure? salt bridge disulfide bridge hydrogen bond hydrophobic interaction
Answers: 1

Chemistry, 22.06.2019 16:00
What rule is used to determine how many covalent bonds an element can form? a. the number of covalent bonds is equal to six c the number of covalent bonds is equal to five minus the group number plus the group number b. the number of covalent bonds is equal to eight d. none of the above minus the group number select the best answer from the choices provided
Answers: 2

Chemistry, 22.06.2019 21:30
The solid xy decomposes into gaseous x and y: xy(s) m x(g) + y(g) kp = 4.1 (at 0 °c) if the reaction is carried out in a 22.4 l container, which initial amounts of x and y will result in the formation of solid xy?
Answers: 1

You know the right answer?
Consider mixture C, which will cause the net reaction to proceed in reverse. Concentration (M)initia...
Questions




Mathematics, 24.02.2021 06:10




History, 24.02.2021 06:10

Mathematics, 24.02.2021 06:10

English, 24.02.2021 06:10

History, 24.02.2021 06:10


History, 24.02.2021 06:10

Mathematics, 24.02.2021 06:10




English, 24.02.2021 06:10

Mathematics, 24.02.2021 06:10

History, 24.02.2021 06:10

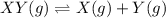
![K_c=\farc{[X][Y]}{[XY]}](/tpl/images/0534/4698/91cd9.png)