
Chemistry, 04.03.2020 23:43 angel213326
Magnesium (used in the manufacture of light alloys) reacts with iron(III) chloride to form magnesium chloride and iron. A mixture of 41.0 g of magnesium and 175.0 g of iron(III) chloride is allowed to react. Identify the limiting reactant and determine the mass of the excess reactant present in the vessel when the reaction is complete.

Answers: 3


Another question on Chemistry


Chemistry, 22.06.2019 15:20
An alloy contains 66 g of pure zinc. what is the percentage of zinc in the alloy? express your answer to two significant figures and include the appropriate units.
Answers: 1

Chemistry, 23.06.2019 01:30
The biomedical technique in which a part of the brain is destroyed with electric current is known as a. electroconvulsive therapy b. prefrontal lobotomy c. bilateral cingulotomy d. tardive dyskinesia
Answers: 2

Chemistry, 23.06.2019 02:00
Calculate the molarity of each aqueous solution: a. 78.0 ml of 0.240 m naoh diluted to 0.250 l with water b. 38.5 ml of 1.2 m hno3 diluted to 0.130 l with water
Answers: 1
You know the right answer?
Magnesium (used in the manufacture of light alloys) reacts with iron(III) chloride to form magnesium...
Questions

Business, 20.08.2019 20:30

English, 20.08.2019 20:30


Mathematics, 20.08.2019 20:30

Social Studies, 20.08.2019 20:30

English, 20.08.2019 20:30


Social Studies, 20.08.2019 20:30

Biology, 20.08.2019 20:30

Mathematics, 20.08.2019 20:30

Mathematics, 20.08.2019 20:30


Mathematics, 20.08.2019 20:30

Mathematics, 20.08.2019 20:30



Mathematics, 20.08.2019 20:30

History, 20.08.2019 20:30


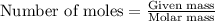 .....(1)
.....(1)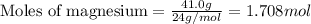
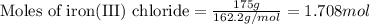
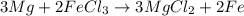
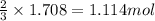 of iron(III) chloride
of iron(III) chloride


