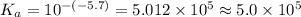Chemistry, 04.03.2020 22:25 payshencec21
The pKa of an acid (one dissociable hydrogen) is -5.7. To the nearest ones, what is the Ka of this acid? Please note that we should use scientific notation and fewer significant figures, but Canvas is not configured to use scientific notation.

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 20:20
Nitric acid can be formed in two steps from the atmospheric gases nitrogen and oxygen, plus hydrogen prepared by reforming natural gas. in the first step, nitrogen and hydrogen react to form ammonia: (g) (g) (g) in the second step, ammonia and oxygen react to form nitric acid and water: (g) (g) (g) (g) calculate the net change in enthalpy for the formation of one mole of nitric acid from nitrogen, hydrogen and oxygen from these reactions. round your answer to the nearest .
Answers: 3

Chemistry, 22.06.2019 23:30
If maltose undergoes hydrolysis what subunits does it results to?
Answers: 2

Chemistry, 23.06.2019 07:30
Type the letter that represents the correct location for each particle type below. the neutron is found at __ the electron is found at __ the proton is found at __
Answers: 1

Chemistry, 23.06.2019 10:30
If a 20.0ml test tube measures 15.0cm, what is the length in meters?
Answers: 1
You know the right answer?
The pKa of an acid (one dissociable hydrogen) is -5.7. To the nearest ones, what is the Ka of this a...
Questions









English, 15.07.2021 18:50

Biology, 15.07.2021 18:50


English, 15.07.2021 18:50




English, 15.07.2021 18:50

Mathematics, 15.07.2021 18:50

Mathematics, 15.07.2021 18:50


 .
.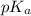 of an acid = -5.7
of an acid = -5.7
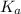 is given by ;
is given by ;![pK_a=-\log[K_a]](/tpl/images/0533/7926/78bbf.png)
![-5.7=-\log[K_a]](/tpl/images/0533/7926/9c3af.png)