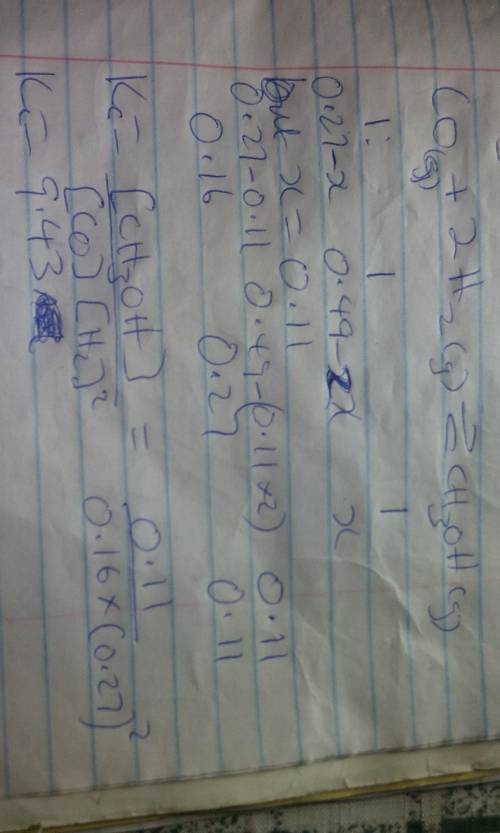Consider the following reaction:
CO(g)+2H2(g)⇌CH3OH(g)
This reaction is carried out at a specific temperature with initial concentrations of [CO] = 0.27 M and [H2] = 0.49 M. At equilibrium, the concentration of CH3OH is 0.11 M. Find the equilibrium constant at this temperature.

Answers: 2


Another question on Chemistry


Chemistry, 22.06.2019 10:30
Astudent reacts 13 moles of iron with 21 moles of oxygen according to the following equation:
Answers: 2

Chemistry, 22.06.2019 19:00
Mercury metal is poured into a graduated cylinder that holds exactly 22.5 ml the mercury used to fill the cylinder mass in 306.0 g from this information calculate the density of mercury
Answers: 2

Chemistry, 22.06.2019 20:30
A40 kilogram skier starts at the top of a 12 meter high slope. at the bottom, she is travelling 10 meters per second. how much energy does she lose to friction
Answers: 2
You know the right answer?
Consider the following reaction:
CO(g)+2H2(g)⇌CH3OH(g)
This reaction is carr...
CO(g)+2H2(g)⇌CH3OH(g)
This reaction is carr...
Questions




History, 08.12.2021 21:50




History, 08.12.2021 21:50

Mathematics, 08.12.2021 21:50

Social Studies, 08.12.2021 21:50

Advanced Placement (AP), 08.12.2021 21:50

English, 08.12.2021 21:50



Social Studies, 08.12.2021 21:50





Chemistry, 08.12.2021 21:50