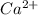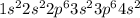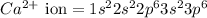Chemistry, 03.03.2020 19:51 Michael845313
The following Lewis diagram represents the valence electron configuration of a main-group element. This element is in group 2A According to the octet rule, this element would be expected to form a(n) with a charge of cation anion If X is in period 4, the ion formed has the same electron configuration as the noble gas The symbol for the ion is

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 09:00
Chemical energy is a form of a. kinetic energy only. b. both potential and kinetic energy. c. neither potential nor kinetic energy. d. potential energy only. reset
Answers: 1

Chemistry, 22.06.2019 11:30
Voltaic cells produce a positive overall charge. what does this indicate? a. the reaction is likely to be endothermic. b. the reaction is spontaneous. c. the reaction is not likely to occur. d. the reaction is not spontaneous.
Answers: 3

Chemistry, 22.06.2019 19:30
Describe the forces both attractive and repulsive that occur as two atoms move closer together.
Answers: 1

Chemistry, 22.06.2019 20:00
The volume of a single vanadium atom is 9.29×10-24 cm3. what is the volume of a vanadium atom in microliters?
Answers: 3
You know the right answer?
The following Lewis diagram represents the valence electron configuration of a main-group element. T...
Questions




Mathematics, 06.03.2021 09:00

Mathematics, 06.03.2021 09:00



Mathematics, 06.03.2021 09:00


Arts, 06.03.2021 09:00

Mathematics, 06.03.2021 09:00


Advanced Placement (AP), 06.03.2021 09:00

Advanced Placement (AP), 06.03.2021 09:00

Health, 06.03.2021 09:00


History, 06.03.2021 09:00


Mathematics, 06.03.2021 09:00

Mathematics, 06.03.2021 09:00