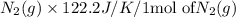Chemistry, 03.03.2020 06:05 Frenchfries13
Consider the reaction: N2(g) + 2O2(g)2NO2(g) Using standard absolute entropies at 298K, calculate the entropy change for the system when 1.90 moles of N2(g) react at standard conditions. S°system = J/K Submit Answer

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 01:00
How can you use chemical equations to predict the products of the reaction you can carry out?
Answers: 1

Chemistry, 22.06.2019 08:30
What are the first three quantum numbers for the electrons located in subshell 2s?
Answers: 2

Chemistry, 22.06.2019 16:00
How will the volume of a gas be affected if the pressure is tripled, but the temperature remains the same?
Answers: 3

Chemistry, 22.06.2019 21:30
How many liters of 3.0 m naoh solution will react with 0.60 liters of 4.0 m h2so4? h2so4 + naoh → na2so4 + h2o 1.2 l 1.6 l 2.4 l 2.8 l
Answers: 3
You know the right answer?
Consider the reaction: N2(g) + 2O2(g)2NO2(g) Using standard absolute entropies at 298K, calculate th...
Questions




History, 31.01.2020 04:45


English, 31.01.2020 04:45

Computers and Technology, 31.01.2020 04:45

Mathematics, 31.01.2020 04:45

Computers and Technology, 31.01.2020 04:45

History, 31.01.2020 04:45

Health, 31.01.2020 04:45




Business, 31.01.2020 04:45




Social Studies, 31.01.2020 04:45

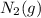 is 191.6 J/mol K,
is 191.6 J/mol K, 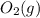 = 205 J/mol K, and
= 205 J/mol K, and 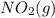 is 239.7 J/mol K at 298 K.
is 239.7 J/mol K at 298 K.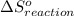 from standard absolute entropies as follows.
from standard absolute entropies as follows.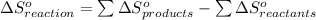
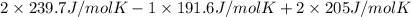
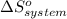 = 1.90 moles of
= 1.90 moles of