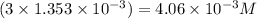Chemistry, 03.03.2020 06:10 andrew8228
A chemist prepares a solution of vanadium(III) bromide (VBr) by measuring out 0.12 g of VBr into a 300 ml. volumetric flask and filling to the mark with distilled water. Calculate the molarity of Branions in the chemist's solution. Be sure your answer is rounded to significant digits.

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 18:10
The enthalpy of formation of water is -285.8 kj/mol. what can be inferred from this statement?
Answers: 1

Chemistry, 21.06.2019 19:30
If the root word engage means “to connect with something,” what does the word disengage mean in the following sentence? he disengaged the gears by stepping on the clutch pedal.a.added more engine powerb.activated a connection to the pedalc.stalled the engined.released a connection to the pedal
Answers: 1

Chemistry, 21.06.2019 22:10
Here’s one way to follow the scientific method. place the missing steps in the correct position in the process
Answers: 1

Chemistry, 22.06.2019 11:30
For each of the following compounds, decide whether the compound's solubility in aqueous solution changes with ph. if the solubility does change, pick the ph at which you'd expect the highest solubility. you'll find ksp data in the aleks data tab. compounds does solubility change with ph
Answers: 3
You know the right answer?
A chemist prepares a solution of vanadium(III) bromide (VBr) by measuring out 0.12 g of VBr into a 3...
Questions

History, 13.07.2019 12:00

History, 13.07.2019 12:00


Biology, 13.07.2019 12:00


Mathematics, 13.07.2019 12:00




Mathematics, 13.07.2019 12:00

Mathematics, 13.07.2019 12:00

Mathematics, 13.07.2019 12:00

Mathematics, 13.07.2019 12:00

Geography, 13.07.2019 12:00

Biology, 13.07.2019 12:00

Biology, 13.07.2019 12:00


History, 13.07.2019 12:00

Chemistry, 13.07.2019 12:00

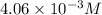
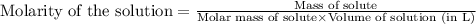
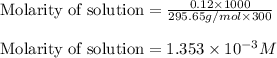
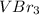
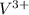 ions and 3 moles of
ions and 3 moles of 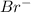 ions
ions