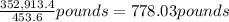Chemistry, 03.03.2020 05:43 freshysans4
A metallurgical firm wishes to dispose of 1200 gallons of waste sulfuric acid whose molarity is 1.05 M. Before disposal, it will be reacted with calcium hydroxide (slaked lime), which costs $0.25 per pound. Write the balanced chemical equation for this process. (Use the lowest possible coefficients. Use the pull-down boxes to specify states such as (aq) or (s). If a box is not needed, leave it blank.) Determine the cost that the firm will incur from this use of slaked lime. Cost

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 19:30
Determine the number o moles of ions/atoms/particle in the following: 2.50 miles of k2s (let me know how to do)
Answers: 1

Chemistry, 21.06.2019 20:30
Pbco3 –> pbo+ co2. how many liters of carbon dioxide gas is produced from the decomposition of 32 grams of lead (ll) carbonate?
Answers: 1

Chemistry, 22.06.2019 13:30
Why does asexual reproduction result in offspring with identicle genetic variation
Answers: 2

Chemistry, 22.06.2019 21:40
A5 mole sample of liquid acetone is converted to a gas at 75.0°c. if 628 j are required to raise the temperature of the liquid to the boiling point, 15.600 kj are required to evaporate the liquid, and 712 j are required to raise the final temperature to 75.0°c, what is the total energy required for the conversion?
Answers: 3
You know the right answer?
A metallurgical firm wishes to dispose of 1200 gallons of waste sulfuric acid whose molarity is 1.05...
Questions


Advanced Placement (AP), 02.04.2020 23:51



Mathematics, 02.04.2020 23:52

Mathematics, 02.04.2020 23:52


Mathematics, 02.04.2020 23:52

English, 02.04.2020 23:52



History, 02.04.2020 23:52


Mathematics, 02.04.2020 23:53

Mathematics, 02.04.2020 23:53



History, 02.04.2020 23:53

English, 02.04.2020 23:53

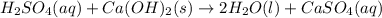
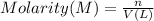
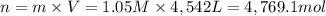
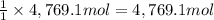 of calcium hydroxide
of calcium hydroxide