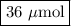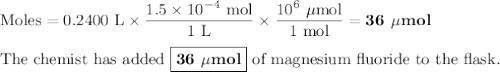Chemistry, 03.03.2020 03:26 ItzAquaZ8716
A chemist adds 240.0mL of a 1.5 x 10^-4 mol/L magnesium flouride (MgF2) solution to a reaction flask. Calculate the micromoles of magnesium fluoride the chemist has added to the flask. Be sure your answer has the correct number of significant digits.

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 12:00
Hey guys so i need to know what is _nh3+> nh4oh ~chemistry~
Answers: 1


Chemistry, 22.06.2019 15:30
Two metal blocks that have slightly different temperatures are placed next to one another. after five minutes, they both have lower but equal temperatures. according to the law of conservation of energy, what most likelyhappened? energy was created inside the blocks.energy was destroyed inside the blocks.energy was absorbed into the blocks from outside the system.energy was transferred from the warmer block to the cooler block.
Answers: 2

Chemistry, 22.06.2019 23:00
In which region is the substance in both the solid phase and the liquid phase? 1 2. 3 4 mark this and return save and exit next
Answers: 2
You know the right answer?
A chemist adds 240.0mL of a 1.5 x 10^-4 mol/L magnesium flouride (MgF2) solution to a reaction flask...
Questions



English, 01.06.2020 02:59


Biology, 01.06.2020 02:59

Mathematics, 01.06.2020 02:59



Health, 01.06.2020 02:59

Physics, 01.06.2020 02:59




Mathematics, 01.06.2020 03:57

Mathematics, 01.06.2020 03:57