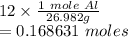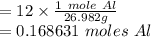Chemistry, 03.03.2020 01:01 darriennichole
Given 12 g of aluminum, how many moles do you have?

Answers: 2


Another question on Chemistry


Chemistry, 22.06.2019 19:20
For a research project, a student decided to test the effect of the lead(ii) ion (pb2+) on the ability of salmon eggs to hatch. this ion was obtainable from the water‐soluble salt, lead(ii) nitrate, which the student decided to make by the following reaction. pbo(s) + 2 hno3(aq) → pb(no3)2(aq) + h2o losses of product for various reasons were expected, and a yield of 86.0% was expected. in order to have 5.00 g of product at this yield, how many grams of pbo should be reacted? (assume that sufficient nitric acid, hno3, would be used.)
Answers: 1

Chemistry, 22.06.2019 23:30
The ammonia molecule in the diagram has the observed bond orientation because
Answers: 1

Chemistry, 23.06.2019 06:30
An engineer decides to use a slightly weaker material rather than a stronger material, since she knows that the stronger material can break suddenly. this is an example of what? a choosing a material that will show warning before it fails b using composite materials that combine strength c using a material for multiple applications d using design techniques that increase efficiency and reduce cost
Answers: 3
You know the right answer?
Given 12 g of aluminum, how many moles do you have?...
Questions


Physics, 05.11.2020 21:40

History, 05.11.2020 21:40

Health, 05.11.2020 21:40





Health, 05.11.2020 21:40

Mathematics, 05.11.2020 21:40

Physics, 05.11.2020 21:40

Mathematics, 05.11.2020 21:40


Chemistry, 05.11.2020 21:40




English, 05.11.2020 21:40

Mathematics, 05.11.2020 21:40

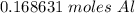
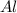 is given as
is given as 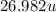 , where
, where  is the unified atomic mass unit.
is the unified atomic mass unit.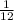 th the mass of a neutral carbon-12 atom. This is equal to the mass of
th the mass of a neutral carbon-12 atom. This is equal to the mass of 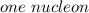 . Therefore:-
. Therefore:-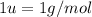
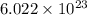 atoms of any element.
atoms of any element.