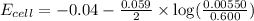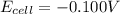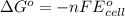Chemistry, 03.03.2020 00:06 cheyennebatz3609
Calculate the emf for the following reaction. Will the reaction occur spontaneously at 25°C, given that [Fe2+] = 0.600 M and [Cd2+] = 0.00550 M? Cd(s) + Fe2+(aq)→Cd2+(aq) + Fe(s) E o Cd2+/Cd = −0.40 V E o Fe2+/Fe = −0.44 V

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 13:30
Aforce of attraction/repulsion due to the spin of electrons what is this force?
Answers: 2

Chemistry, 21.06.2019 20:30
9. write the chemical equation for the following word equations. include symbols for physical states in the equation. a. solid zinc sulfide + oxygen gas -> solid zinc oxide + sulfur dioxide gas b. aqueous hydrochloric acid + aqueous barium hydroxide -> aqueous barium chloride + water
Answers: 1

Chemistry, 22.06.2019 03:10
Agas diffuses 1/7 times faster than hydrogen gas (h2). what is the molar mass of the gas? 100.10 g/mol 98.78 g/mol 86.68 g/mol 79.98 g/mol
Answers: 3

You know the right answer?
Calculate the emf for the following reaction. Will the reaction occur spontaneously at 25°C, given t...
Questions


Social Studies, 10.10.2019 21:00

Mathematics, 10.10.2019 21:00

Chemistry, 10.10.2019 21:00

Health, 10.10.2019 21:00


Biology, 10.10.2019 21:00






Mathematics, 10.10.2019 21:00



Mathematics, 10.10.2019 21:00

Mathematics, 10.10.2019 21:00



Mathematics, 10.10.2019 21:00

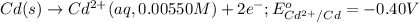
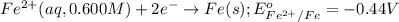
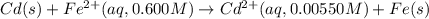
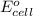 of the reaction, we use the equation:
of the reaction, we use the equation: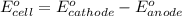
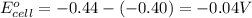
![E_{cell}=E^o_{cell}-\frac{0.059}{n}\log \frac{[Cd^{2+}]}{[Fe^{2+}]}](/tpl/images/0531/1947/ee65e.png)
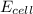 = electrode potential of the cell = ?
= electrode potential of the cell = ?![[Cd^{2+}]=0.00550M](/tpl/images/0531/1947/3e0ad.png)
![[Fe^{2+}]=0.600M](/tpl/images/0531/1947/5e50b.png)