
Chemistry, 03.03.2020 00:07 grayfaith16
Consider the phase change of ice to liquid water if the molar heat of fusion for water is 6.01 kj mol-1 how many joules are required to melt 1 gram of ice at 273 k

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 18:00
Which type of reaction always has an element and a compound as reactants
Answers: 1

Chemistry, 22.06.2019 00:30
What is the most stable monatomic ion formed from nitrogen
Answers: 2

Chemistry, 22.06.2019 02:00
For each of the following types of reactions, write a general reaction formula in the symbolic form—for example, a + b → ab. single-displacement double-displacement synthesis decomposition
Answers: 1

Chemistry, 22.06.2019 11:40
Which type of precipitation would most likely form when the surface air temperature is slightly below freezing and the air temperature increases as you move upward away from the ground?
Answers: 2
You know the right answer?
Consider the phase change of ice to liquid water if the molar heat of fusion for water is 6.01 kj mo...
Questions



Mathematics, 03.10.2021 17:10

Physics, 03.10.2021 17:10

Biology, 03.10.2021 17:10



Biology, 03.10.2021 17:10


Mathematics, 03.10.2021 17:10



English, 03.10.2021 17:10

History, 03.10.2021 17:10


History, 03.10.2021 17:10

Mathematics, 03.10.2021 17:10

Mathematics, 03.10.2021 17:10


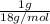 = 0.055 mol
= 0.055 mol

