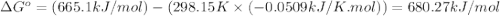Chemistry, 02.03.2020 21:22 jocelynmarquillo1
"Thermite" reactions have been used for welding metal parts such as railway rails and in metal refining. One such thermite reaction is 3 Mg(s) + Cr2O3(s) → 3 MgO(s) + 2 Cr(s). During the reaction, the surroundings absorb 665.1 kJ/mol of heat. Is the reaction spontaneous at room temperature (298.15 K) under standard conditions?

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 08:00
This classification of drug typically changes the brain's chemistry and reduces its ability to create its own endorphins.
Answers: 1

Chemistry, 22.06.2019 12:40
When 13.3 g koh is dissolved in 102.7 g of water in a coffee-cup calorimeter, the temperature rises from 21.4 °c to 31.53 °c. what is the enthalpy change per gram of koh (j/g) dissolved in the water? * take the density of water as 1.00 g/ml. * assume that the solution has a specific heat capacity of 4.18 j/g*k. enter to 1 decimal place. do not forget the appropriate sign /(+). canvas may auto-delete the (+) sign
Answers: 2

Chemistry, 22.06.2019 13:00
Is 9 correct? and can someone me with 10? it’s due tomorrow, you
Answers: 1

Chemistry, 22.06.2019 14:30
Need ! asap will mark 10 pts using the room temperature line (orange line) and your periodic table, make lists that identify the state of matter (gas, liquid, or solid) in which each element you plotted exists at room temperature. explain your answers
Answers: 1
You know the right answer?
"Thermite" reactions have been used for welding metal parts such as railway rails and in metal refin...
Questions

History, 19.07.2019 19:50



Mathematics, 19.07.2019 20:00


Mathematics, 19.07.2019 20:00




Business, 19.07.2019 20:00


Mathematics, 19.07.2019 20:00



History, 19.07.2019 20:00





![\Delta S^o_{rxn}=\sum [n\times \Delta S^o_{(product)}]-\sum [n\times \Delta S^o_{(reactant)}]](/tpl/images/0530/7449/52737.png)
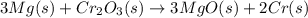
![\Delta S^o_{rxn}=[(3\times \Delta S^o_{(MgO(s))})+(2\times \Delta S^o_{(Cr(s))})]-[(3\times \Delta S^o_{(Mg(s))})+(1\times \Delta S^o_{(Cr_2O_3(s))})]](/tpl/images/0530/7449/80d1e.png)
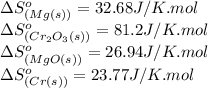
![\Delta S^o_{rxn}=[(3\times (26.94))+(2\times (23.77))]-[(3\times (32.68))+(1\times (81.2))]\\\\\Delta S^o_{rxn}=-50.88J/K=-0.0509kJ/K.mol](/tpl/images/0530/7449/fa231.png)
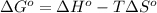
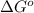 = standard Gibbs free energy = ?
= standard Gibbs free energy = ?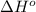 = standard enthalpy change of the reaction = 665.1 kJ/mol
= standard enthalpy change of the reaction = 665.1 kJ/mol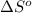 = standard entropy change of the reaction = -0.0509 kJ/K.mol
= standard entropy change of the reaction = -0.0509 kJ/K.mol