
Chemistry, 02.03.2020 19:12 chandranewlon
The student determines the molar mass of the gas to be 64 g mol-1. write the expression (set-up) for calculating the percent error in the experimental value, assuming that the unknown gas is butane (molar mass 58 g mol-1). calculations are not required

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 21:00
Agas in a balloon at constant pressure has a volume of 160 ml at -125*c. what is its volume at 29.0*c?
Answers: 1

Chemistry, 22.06.2019 07:00
The organism shown is a free-living one that is anchored to the bottom of ponds and streams during one stage of its life cycle what is the common name for the group to which this organism belong
Answers: 3

Chemistry, 22.06.2019 16:30
At 20°c, a sample of h2o liquid and a sample of co2 gas each have the same average kinetic energy. why is one a liquid and the other a gas at this temperature?
Answers: 1

Chemistry, 22.06.2019 18:00
The fact that the total amount of energy in a system remains constant is a(n)
Answers: 1
You know the right answer?
The student determines the molar mass of the gas to be 64 g mol-1. write the expression (set-up) for...
Questions

Mathematics, 28.01.2020 05:31

Mathematics, 28.01.2020 05:31

Mathematics, 28.01.2020 05:31


Advanced Placement (AP), 28.01.2020 05:31

Mathematics, 28.01.2020 05:31


History, 28.01.2020 05:31

English, 28.01.2020 05:31

Chemistry, 28.01.2020 05:31


Mathematics, 28.01.2020 05:31

English, 28.01.2020 05:31


English, 28.01.2020 05:31



Mathematics, 28.01.2020 05:31


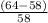 x 100%
x 100%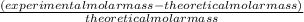 x 100%
x 100%

