Chemistry, 02.03.2020 17:55 shawnr6989
Calculate the amount of heat required to convert 10.0 grams of ice at –20.°C to steam at 120.°C. (Sp. heat of H2O(s) = 2.09 J/g•°C, Sp. heat of H2O(l) = 4.18 J/g•°C, Sp heat of H2O(g) = 2.03 J/g•°C; heat of fus. of H2O(s) = 333 J/g, heat of vap. of H2O(l) = 2260 J/g).

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 10:50
How many grams of oxygen gas are contained in a 15 l sample at 1.02 atm and 28°c? show your work.
Answers: 1


Chemistry, 23.06.2019 01:30
If a particle has z = 25 and 23 electrons, what is its charge?
Answers: 2

Chemistry, 23.06.2019 03:30
The semi-conductors on the periodic table are classified as
Answers: 1
You know the right answer?
Calculate the amount of heat required to convert 10.0 grams of ice at –20.°C to steam at 120.°C. (Sp...
Questions


History, 30.01.2021 01:00


Mathematics, 30.01.2021 01:00

History, 30.01.2021 01:00

Advanced Placement (AP), 30.01.2021 01:00





English, 30.01.2021 01:00

Mathematics, 30.01.2021 01:00



English, 30.01.2021 01:00

Mathematics, 30.01.2021 01:00

Mathematics, 30.01.2021 01:00



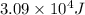
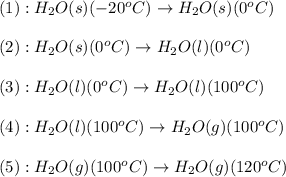
![\Delta H=[m\times c_{p,s}\times (T_{final}-T_{initial})]+m\times \Delta H_{fusion}+[m\times c_{p,l}\times (T_{final}-T_{initial})]+m\times \Delta H_{vap}+[m\times c_{p,g}\times (T_{final}-T_{initial})]](/tpl/images/0530/2584/4a4bb.png)
 = heat required for the reaction
= heat required for the reaction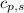 = specific heat of solid water or ice =
= specific heat of solid water or ice = 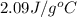
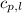 = specific heat of liquid water =
= specific heat of liquid water = 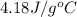
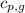 = specific heat of gaseous water =
= specific heat of gaseous water = 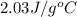
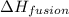 = enthalpy change for fusion =
= enthalpy change for fusion = 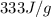
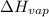 = enthalpy change for vaporization =
= enthalpy change for vaporization = 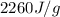
![\Delta H=[10.0g\times 2.09J/g^oC\times (0-(-20))^oC]+10.0g\times 333J/g+[10.0g\times 4.18J/g^oC\times (100-0)^oC]+10.0g\times 2260J/g+[10.0g\times 2.03J/g^oC\times (120-100)^oC]](/tpl/images/0530/2584/295b1.png)