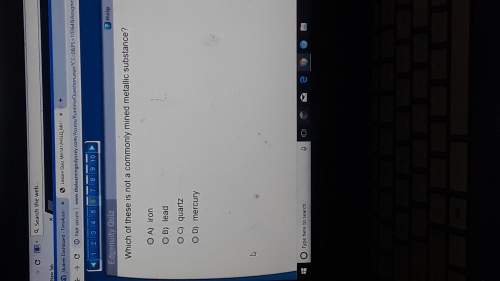
Select all that apply.
values of ksp greater than 1.0 infer:
the solid phase is relat...

Chemistry, 23.08.2019 03:40 carladasie
Select all that apply.
values of ksp greater than 1.0 infer:
the solid phase is relatively insoluble.
the solid phase is relatively soluble.
the chemical reaction proceeds readily to the right.
the chemical reaction proceeds readily to the left.

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 06:10
Explain the relationship between forward and backward reactions in equilibrium, and predict how changing the amount of a reactant (creating a tension) will affect that relationship.
Answers: 1

Chemistry, 22.06.2019 20:00
Many free radicals combine to form molecules that do not contain any unpaired electrons. the driving force for the radical–radical combination reaction is the formation of a new electron‑pair bond. consider the chemical equation. n(g)+no(g)⟶nno(g) n(g)+no(g)⟶nno(g) write lewis formulas for the reactant and product species in the chemical equation. include nonbonding electrons. n(g)n(g) select draw rings more erase select draw rings more erase select draw rings more erase n no(g)
Answers: 1


Chemistry, 23.06.2019 04:00
What changes occur in the reaction indicated by the equation? check all that apply. the hydrogen nucleus loses protons. the oxygen nucleus gains protons. the bond in h2 is broken, and new bonds are formed between hydrogen and oxygen atoms. each electron associated with a hydrogen atom is shared with an oxygen atom.
Answers: 3
You know the right answer?
Questions


Mathematics, 17.01.2022 01:00

Mathematics, 17.01.2022 01:00

Physics, 17.01.2022 01:00


History, 17.01.2022 01:00

Mathematics, 17.01.2022 01:00

English, 17.01.2022 01:00


Computers and Technology, 17.01.2022 01:00

Mathematics, 17.01.2022 01:00

Mathematics, 17.01.2022 01:00

Mathematics, 17.01.2022 01:00

Mathematics, 17.01.2022 01:00




SAT, 17.01.2022 01:00

History, 17.01.2022 01:00

Health, 17.01.2022 01:00