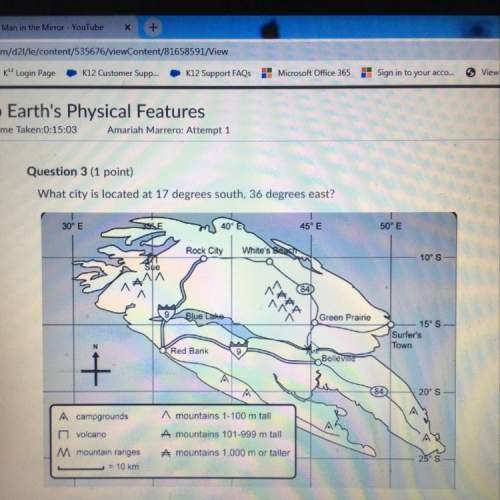
Chemistry, 29.02.2020 03:29 gisellekarime
Aqueous potassium phosphate reacts with aqueous aluminum chloride to form aqueous potassium chloride and solid aluminum phosphate. Write the balanced chemical equation for the reaction. Include physical states.

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 13:30
Table sugar completely dissolved in water is an example of a?
Answers: 1

Chemistry, 22.06.2019 15:00
Large helium-filled balloons are used to lift scientific equipment to high altitudes. what is the pressure inside such a balloon if it starts out at sea level with a temperature of 10.0ºc and rises to an altitude where its volume is twenty times the original volume and its temperature is – 50.0ºc ?
Answers: 2

Chemistry, 22.06.2019 21:50
Answer the questions about this reaction: nai(aq) + cl2(g) → nacl(aq) + i2(g) write the oxidation and reduction half-reactions: oxidation half-reaction: reduction half-reaction: based on the table of relative strengths of oxidizing and reducing agents (b-18), would these reactants form these products? write the balanced equation: answer options: a. 0/na -> +1/na+1e- b. nai(aq) + cl2(g) → nacl(aq) + i2(g) c. +1/na+1e- -> 0 /na d. -1/2i -> 0/i2+2e- e. no f. 4nai(aq) + cl2(g) → 4nacl(aq) + i2(g) g. 2nai(aq) + cl2(g) → 2nacl(aq) + i2(g) h. 4nai(aq) + 2cl2(g) → 4nacl(aq) + 2i2(g) i. nai(aq) + cl2(g) → nacl(aq) + i2(g) j. 0/cl2+2e -> -1/2cl- k. yes
Answers: 1

Chemistry, 22.06.2019 22:30
Which is a characteristic of the electron sea model for metallic bonding? molecular orbitals overlap to produce bands. electrons flow easily between metal nuclei. electrons are in fixed positions in the orbitals. atomic nuclei are arranged in an irregular pattern.
Answers: 3
You know the right answer?
Aqueous potassium phosphate reacts with aqueous aluminum chloride to form aqueous potassium chloride...
Questions

History, 09.07.2019 11:30

History, 09.07.2019 11:30

Computers and Technology, 09.07.2019 11:30



Geography, 09.07.2019 11:30

History, 09.07.2019 11:30


English, 09.07.2019 11:30

History, 09.07.2019 11:30


History, 09.07.2019 11:30


Mathematics, 09.07.2019 11:30

Social Studies, 09.07.2019 11:30

Chemistry, 09.07.2019 11:30

English, 09.07.2019 11:30


Biology, 09.07.2019 11:30

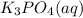 +
+ 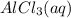 --->
---> 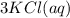 +
+