
Chemistry, 28.02.2020 20:06 michellen2020
The first two steps in the industrial synthesis of nitric acid produce nitrogen dioxide from ammonia: 2N0(g) +02(g) 2NO2 (g) The net reaction is: 4NH, (g) [email protected])= 4 Write an equation that gives the overall equilibrium constant K in terms of the equilibrium constants K1 and K2. If you need to include any physical constants, be sure you use their standard symbols, which you'll find in the ALEKS Calculator. K=

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 20:30
As you move from right to left on the periodic table the atomic radius fill in the blank
Answers: 2

Chemistry, 22.06.2019 21:30
The solid xy decomposes into gaseous x and y: xy(s) m x(g) + y(g) kp = 4.1 (at 0 °c) if the reaction is carried out in a 22.4 l container, which initial amounts of x and y will result in the formation of solid xy?
Answers: 1

Chemistry, 22.06.2019 21:30
How many oxygen atoms are there in 3.15 moles of hcl manganese (iv) oxide, mno2
Answers: 2

Chemistry, 23.06.2019 09:20
1) a. water molecule breaks up into hydrogen and oxygen on passing electricity. does this involve breaking intermolecular or intramolecular forces of attraction. explain b. on boiling water changes to water vapor. does this involve breaking intermolecular or intramolecular forces of attraction. explain methanol evaporates faster than water. contrast the intermolecular forces and the vapor pressures of methanol and water?
Answers: 2
You know the right answer?
The first two steps in the industrial synthesis of nitric acid produce nitrogen dioxide from ammonia...
Questions




English, 03.02.2020 23:02

Social Studies, 03.02.2020 23:02


Mathematics, 03.02.2020 23:03

Biology, 03.02.2020 23:03

Mathematics, 03.02.2020 23:03

Mathematics, 03.02.2020 23:03


Chemistry, 03.02.2020 23:03

Computers and Technology, 03.02.2020 23:03

History, 03.02.2020 23:03





Chemistry, 03.02.2020 23:03

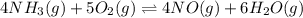
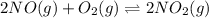
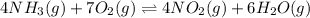
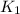 and
and 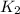 . If you need to include any physical constants, be sure you use their standard symbols
. If you need to include any physical constants, be sure you use their standard symbols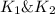 :
: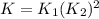
![K_1=\frac{[NO]^4[H_2O]^6}{[NH_3]^4[O_2]^5}](/tpl/images/0528/3548/6d44f.png)
![K_2=\frac{[NO_2]^2}{[NO]^2[O_2]}](/tpl/images/0528/3548/04922.png)
![K=\frac{[NO_2]^4[H_2O]^6}{[NH_3]^4[O_2]^7}](/tpl/images/0528/3548/d6ea1.png)
![[NO]^4](/tpl/images/0528/3548/e5e10.png) ;
;![K=\frac{[NO_2]^4[H_2O]^6}{[NH_3]^4[O_2]^7}\times \frac{[NO]^4}{[NO]^4}](/tpl/images/0528/3548/cc872.png)
![K=\frac{[NO]^4[H_2O]^6}{[NH_3]^4[O_2]^7}\times \frac{[NO_2]^4}{[NO]^4}](/tpl/images/0528/3548/b9d44.png)
![K=K_1\times \frac{[NO_2]^4}{[O_2]^2[NO]^4}](/tpl/images/0528/3548/1f7b8.png)
![K=K_1\times (\frac{[NO_2]^2}{[O_2]^1[NO]^2})^2](/tpl/images/0528/3548/d8543.png)


