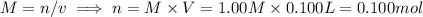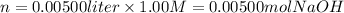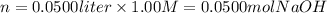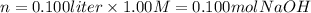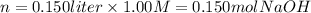Chemistry, 28.02.2020 19:46 robert7248
A) A volume of 100 mL of 1.00 M HCl solution is titrated with 1.00 M NaOH solution. You added the following quantities of 1.00 M NaOH to the reaction flask. Classify the following conditions after the equivalence point. Write down the items to their respective table.
i) 5.00 mL of 1.00 M NaOH
ii) 50.0 mL of 1.00 M NaOH
iii) 100 mL of 1.00 M NaOH
iv) 150 mL of 1.00 M NaOH
v) 200 mL of 1.00 M NaOH
Before equivalence point At equivalence point After equivalence point

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 16:50
Which element is least likely to undergo a chemical reaction
Answers: 3

Chemistry, 22.06.2019 17:00
The msds for glacial acetic acid says that it is a flammable liquid that can severely burn any human tissue it comes in contact with. it reacts with bases, various metals, and strong oxidizing agents. its vapors can form explosive mixtures with air.
Answers: 1

Chemistry, 23.06.2019 01:30
What is the importance of interlocking the fingers and rubbing while washing hands? the palms are the dirtiest parts of the hands. the spaces between the fingers get washed. the backs of the hands get washed. the fingernails are the dirtiest parts of the hands
Answers: 1

Chemistry, 23.06.2019 08:50
What happens after sound waves create vibrations in the ear?
Answers: 1
You know the right answer?
A) A volume of 100 mL of 1.00 M HCl solution is titrated with 1.00 M NaOH solution. You added the fo...
Questions

Mathematics, 09.09.2020 08:01

Mathematics, 09.09.2020 08:01

Mathematics, 09.09.2020 08:01

Mathematics, 09.09.2020 08:01

Mathematics, 09.09.2020 08:01

Mathematics, 09.09.2020 08:01

Mathematics, 09.09.2020 08:01

Mathematics, 09.09.2020 08:01

Mathematics, 09.09.2020 08:01

French, 09.09.2020 08:01

Mathematics, 09.09.2020 08:01

English, 09.09.2020 08:01

Mathematics, 09.09.2020 08:01

English, 09.09.2020 08:01

Mathematics, 09.09.2020 08:01

Mathematics, 09.09.2020 08:01

Mathematics, 09.09.2020 08:01

Mathematics, 09.09.2020 08:01

Mathematics, 09.09.2020 08:01

Mathematics, 09.09.2020 08:01