
Chemistry, 28.08.2019 14:50 meadowsoares7
The balanced chemical equation for the reaction of copper (cu) and silver nitrate (agno3) is shown below.
cu + 2agno3 mc001-1.jpg 2ag + cu(no3)2
how many moles of copper must react to form 3.50 mol of ag?
0.175 mol
1.75 mol
7.00 mol
17.0 mol

Answers: 2


Another question on Chemistry


Chemistry, 22.06.2019 00:40
Base your answer on the information below and on your knowledge of chemistry. nitrogen dioxide, no2, is a dark brown gas that is used to make nitric acid and to bleach flour. nitrogen dioxide has a boiling point of 294 k at 101.3 kpa. in a rigid cylinder with a movable piston, nitrogen dioxide can be in equilibrium with colorless dinitrogen tetroxide, n2o4. this equilibrium is represented by the equation below. 2no2(g) n2o4(g) + 58kj at standard pressure, compare the strength of intermolecular forces in no2(g) to the strength of intermolecular forces in n2(g).
Answers: 2

Chemistry, 22.06.2019 01:00
Which statement correctly describes potassium iodide, ki? there is a one-to-one ratio of potassium ions to iodide ions. potassium gains electrons and iodine loses electrons during the reaction. the lattice is held together by potassium anions and iodide cations.
Answers: 1

Chemistry, 22.06.2019 06:00
What type of electromagnetic radiation has a shorter wavelength than blue light
Answers: 2
You know the right answer?
The balanced chemical equation for the reaction of copper (cu) and silver nitrate (agno3) is shown b...
Questions

History, 05.02.2021 01:00


Mathematics, 05.02.2021 01:00

Biology, 05.02.2021 01:00

Mathematics, 05.02.2021 01:00

Mathematics, 05.02.2021 01:00

Biology, 05.02.2021 01:00



English, 05.02.2021 01:00


Biology, 05.02.2021 01:00

Mathematics, 05.02.2021 01:00

Mathematics, 05.02.2021 01:00


Mathematics, 05.02.2021 01:00

English, 05.02.2021 01:00

History, 05.02.2021 01:00


Mathematics, 05.02.2021 01:00

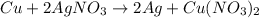
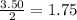 moles of copper
moles of copper

