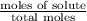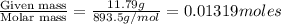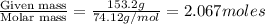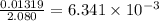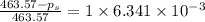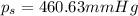Chemistry, 28.02.2020 19:30 samantha636
The vapor pressure of diethyl ether (ether) is 463.57 mm Hg at 25 °C. A nonvolatile, nonelectrolyte that dissolves in diethyl ether is chlorophyll. Calculate the vapor pressure of the solution at 25 °C when 11.79 grams of chlorophyll, C55H72MgN4O5 (893.5 g/mol), are dissolved in 153.2 grams of diethyl ether. diethyl ether = CH3CH2OCH2CH3 = 74.12 g/mol. VP(solution) = mm Hg

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 10:00
How many grams of co(g) are there in 74.5 ml of the gas at 0.933 atm and 30o c?
Answers: 1

Chemistry, 22.06.2019 12:30
According to the valence shell electron pair repulsion (vsepr) theory, a molecule that has four electron groups around the central atom will exhibit what electron geometry? view available hint(s) according to the valence shell electron pair repulsion (vsepr) theory, a molecule that has four electron groups around the central atom will exhibit what electron geometry? trigonal bipyramidal tetrahedral square planar determination of electron geometry requires information on whether the electron groups are lone pairs or bonding groups.
Answers: 2

Chemistry, 23.06.2019 02:30
Which of the four hypothetical substances you investigated would be most harmful to living organisms? 50 points!
Answers: 2

Chemistry, 23.06.2019 08:40
The half-life of a certain element is 100 days. how many half-lives will it be before only one eighth of this elementremains?
Answers: 1
You know the right answer?
The vapor pressure of diethyl ether (ether) is 463.57 mm Hg at 25 °C. A nonvolatile, nonelectrolyte...
Questions

Mathematics, 15.12.2021 18:20



Business, 15.12.2021 18:20

Chemistry, 15.12.2021 18:20



Chemistry, 15.12.2021 18:20


Mathematics, 15.12.2021 18:20

Mathematics, 15.12.2021 18:20

English, 15.12.2021 18:20

Law, 15.12.2021 18:20

Arts, 15.12.2021 18:30



Mathematics, 15.12.2021 18:30

History, 15.12.2021 18:30

Mathematics, 15.12.2021 18:30

Mathematics, 15.12.2021 18:30

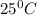 is 460.63 mmHg
is 460.63 mmHg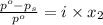
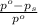 = relative lowering in vapor pressure
= relative lowering in vapor pressure
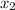 = mole fraction of solute =
= mole fraction of solute =