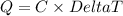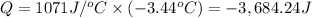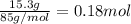Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 13:00
Based on the position vs. time graph which velocity vs. time graph would correspond to the data
Answers: 2

Chemistry, 22.06.2019 02:00
How many moles of magnesium is 3.01 x10^22 atoms of magnesium?
Answers: 1

Chemistry, 22.06.2019 08:40
Write the formula for the following chemicals. 7. e. trinitrogen tetraoxide a calcium phosphate f. magnesium acetate b. potassium sulfide g nickel(iii) cyanide c carbon dioxide h. silver sulfate d. cobalt(ii) chloride
Answers: 1

Chemistry, 22.06.2019 10:50
8) a mixture of he, ne and ar has a pressure of 7.85 atm. if the ne has a mole fraction of 0.47 and 8) ar has a mole fraction of 0.23, what is the pressure of he? a) 4.2 atm b) 3.7 atm c) 5.5 atm d) 2.4 atm e) 1.8 atm
Answers: 1
You know the right answer?
When 15.3 g of sodium nitrate, NaNO3,was dissolved in water in a calorimeter, the temperature fell f...
Questions




Mathematics, 14.07.2020 01:01




Mathematics, 14.07.2020 01:01

Mathematics, 14.07.2020 01:01

Mathematics, 14.07.2020 01:01


Mathematics, 14.07.2020 01:01

Chemistry, 14.07.2020 01:01


Computers and Technology, 14.07.2020 01:01





Biology, 14.07.2020 01:01