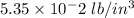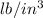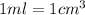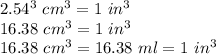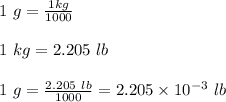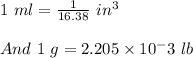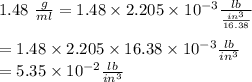Chemistry, 27.02.2020 05:47 faithyholcomb
11. The density of liquid chloroform is 1.48 g/mL. What is its density in units of lb/in?
(2.54 cm = 1 in., 2.205 lb = 1 kg)

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 18:30
150.0 grams of asf3 were reacted with 180.0 g of ccl4 to produce ascl3 and ccl2f2. if the actual yield of ccl2f2 was 127 g, what is the percent yield?
Answers: 2

Chemistry, 22.06.2019 05:00
Choose all the answers that apply. ionic compounds dissolve easily in water do not dissolve in water have low melting points have high melting points conduct electricity when melted
Answers: 1

Chemistry, 22.06.2019 12:40
Consider the directing effects of the substituents on salicylamide and predict the possible structures of the iodination products. which do you think will be the major product?
Answers: 1

Chemistry, 23.06.2019 07:20
Which statement explains which component is likely to be more powerful in explaining a scientific phenomenon? a) component c, because a theory is often passed on possibility and not certainty b) component d, because a hypothesis is often based on possibility not certainty c) component c, because the ability to explain several occurrences in the natural world is a characteristic of a hypothesis d) component d, because the ability to explain several occurrences in the natural world is a characteristic of a theory
Answers: 3
You know the right answer?
11. The density of liquid chloroform is 1.48 g/mL. What is its density in units of lb/in?
Questions


Chemistry, 16.11.2020 14:00

History, 16.11.2020 14:00

Chemistry, 16.11.2020 14:00


Mathematics, 16.11.2020 14:00

Chemistry, 16.11.2020 14:00



Mathematics, 16.11.2020 14:00

Social Studies, 16.11.2020 14:00


History, 16.11.2020 14:00

Chemistry, 16.11.2020 14:00


Mathematics, 16.11.2020 14:00

Mathematics, 16.11.2020 14:00

Mathematics, 16.11.2020 14:00


Social Studies, 16.11.2020 14:00