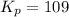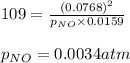The following reaction has Kp = 109 at 25°C. 2 NO(g) + Br2(g) equilibrium reaction arrow 2 NOBr(g) If the equilibrium partial pressure of Br2 is 0.0159 atm and the equilibrium partial pressure of NOBr is 0.0768 atm, calculate the partial pressure of NO at equilibrium.

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 06:00
An atom of lithium (li) and an atom of chlorine (cl) engage in a chemical reaction. which correctly describes the structure of the resulting chemical compound? hint: consider the class of each element. the chemical compound will have a network structure. the chemical compound will have triple bonds. the chemical compound will have a ball-and-stick structure. the chemical compound will have double bonds.
Answers: 2

Chemistry, 22.06.2019 08:30
For each of the compounds below, show that the charges on the ions add up to zero. a. kbr b. cao c. li(2)o d. cacl(2) e. alcl(3)
Answers: 2


Chemistry, 22.06.2019 19:00
Structure of the atoms: discovery of the nucleus in 1909i need answering all of these questions
Answers: 3
You know the right answer?
The following reaction has Kp = 109 at 25°C. 2 NO(g) + Br2(g) equilibrium reaction arrow 2 NOBr(g) I...
Questions

History, 09.10.2019 06:00


Biology, 09.10.2019 06:00



World Languages, 09.10.2019 06:00


History, 09.10.2019 06:00


English, 09.10.2019 06:00


Biology, 09.10.2019 06:00








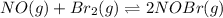
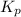 for above equation follows:
for above equation follows: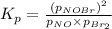
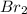 = 0.0159 atm
= 0.0159 atm