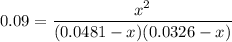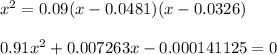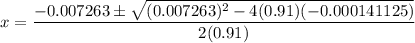Chemistry, 26.02.2020 20:51 jaystitt1152683
At 25°C, K = 0.090 for the following reaction. H2O(g) + Cl2O(g) equilibrium reaction arrow 2 HOCl(g) Calculate the concentrations of all species at equilibrium for each of the following cases. (a) 1.3 g H2O and 2.2 g Cl2O are mixed in a 1.5-L flask.

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 14:40
Water ionizes by the equation h2o(l)⇌h+(aq)+oh−(aq) the extent of the reaction is small in pure water and dilute aqueous solutions. this reaction creates the following relationship between [h+] and [oh−]: kw=[h+][oh−] keep in mind that, like all equilibrium constants, the value of kw changes with temperature.
Answers: 1

Chemistry, 22.06.2019 12:10
Consider the reaction: n2(g) + o2(g) ⇄ 2no(g) kc = 0.10 at 2000oc starting with initial concentrations of 0.040 mol/l of n2 and 0.040 mol/l of o2, calculate the equilibrium concentration of no in mol/l how would this be done?
Answers: 3

Chemistry, 22.06.2019 20:30
Calculate the percent composition by mass of each element in al(oh)3. use at least three significant figures.
Answers: 1

Chemistry, 22.06.2019 21:00
Which answer tells the reason the earth’s climate is getting warmer? too many animals are becoming extinct. large glaciers are melting in antarctica. the earth is moving closer to the sun. driving cars gives off gases that trap heat in the atmosphere.
Answers: 1
You know the right answer?
At 25°C, K = 0.090 for the following reaction. H2O(g) + Cl2O(g) equilibrium reaction arrow 2 HOCl(g)...
Questions

English, 06.12.2019 03:31



History, 06.12.2019 03:31


History, 06.12.2019 03:31

Mathematics, 06.12.2019 03:31


History, 06.12.2019 03:31

Biology, 06.12.2019 03:31



English, 06.12.2019 03:31






Computers and Technology, 06.12.2019 03:31

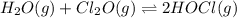
![K_c=\dfrac{[HOCl]^2}{[H_2O].[Cl_2O]}](/tpl/images/0525/3967/fd69c.png)