

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 04:30
How much energy is made when a pice of wood burns. how do you know
Answers: 2

Chemistry, 22.06.2019 16:30
Ammonium perchlorate nh4clo4 is the solid rocket fuel used by the u.s. space shuttle. it reacts with itself to produce nitrogen gas n2 , chlorine gas cl2 , oxygen gas o2 , water h2o , and a great deal of energy. what mass of nitrogen gas is produced by the reaction of 2.1g of ammonium perchlorate?
Answers: 2

Chemistry, 22.06.2019 20:00
Many free radicals combine to form molecules that do not contain any unpaired electrons. the driving force for the radical–radical combination reaction is the formation of a new electron‑pair bond. consider the chemical equation. n(g)+no(g)⟶nno(g) n(g)+no(g)⟶nno(g) write lewis formulas for the reactant and product species in the chemical equation. include nonbonding electrons. n(g)n(g) select draw rings more erase select draw rings more erase select draw rings more erase n no(g)
Answers: 1

Chemistry, 22.06.2019 20:30
Calculate the percent composition by mass of each element in al(oh)3. use at least three significant figures.
Answers: 1
You know the right answer?
A reaction has a rate constant of 2.08 × 10−4 s−1 at 26 oC and 0.394 s−1 at 79 oC . Determine the ac...
Questions



Mathematics, 27.02.2020 21:40


History, 27.02.2020 21:40


Mathematics, 27.02.2020 21:40







Arts, 27.02.2020 21:41

Medicine, 27.02.2020 21:41

Biology, 27.02.2020 21:41



Mathematics, 27.02.2020 21:41

History, 27.02.2020 21:41

![\ln(\frac{K_{79^oC}}{K_{26^oC}})=\frac{E_a}{R}[\frac{1}{T_1}-\frac{1}{T_2}]](/tpl/images/0525/0731/e30ee.png)
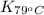 = equilibrium constant at 79°C =
= equilibrium constant at 79°C = 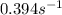
 = equilibrium constant at 26°C =
= equilibrium constant at 26°C = 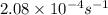
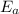 = Activation energy of the reaction = ?
= Activation energy of the reaction = ?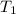 = initial temperature =
= initial temperature = ![26^oC=[26+273]K=299K](/tpl/images/0525/0731/6a452.png)
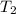 = final temperature =
= final temperature = ![79^oC=[79+273]K=352K](/tpl/images/0525/0731/91aa7.png)
![\ln(\frac{0.394}{2.08\times 10^{-4}})=\frac{E_a}{8.314J/mol.K}[\frac{1}{299}-\frac{1}{352}]\\\\E_a=124595J/mol=124.6kJ/mol](/tpl/images/0525/0731/ebde0.png)


