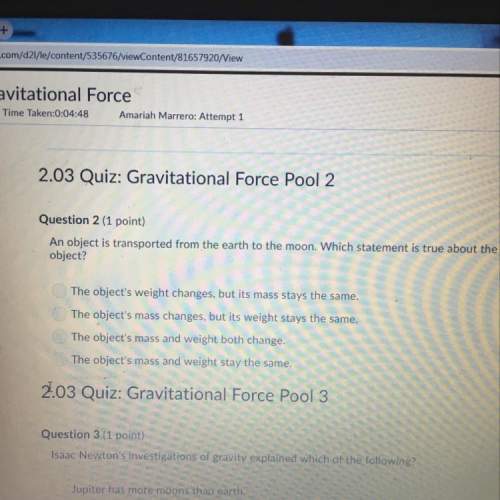
Chemistry, 26.02.2020 07:33 sarahcyberpony
7.0 g of nitrogen is reacted with 5.0 g of hydrogen to produce ammonia according to the chemical equation shown below. Which one of the following statements is false?
N2(g) + 3 H2(g) → 2 NH3(g)

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 15:00
An example of technology is the a. addition of a side group to an organic molecule during synthesis. b. use of a new antibiotic to fight an infection. c. measurement of iron concentration in a water sample. d. study of atomic fusion reactions
Answers: 3

Chemistry, 22.06.2019 15:30
What best discribes the relationship between wavelength and frequency in a electromagnetic wave
Answers: 1

Chemistry, 22.06.2019 15:30
Which statement names the physical property of wood a. wood is softer than coal b. wood does not rust c. wood can rot d. wood can burn
Answers: 1

Chemistry, 22.06.2019 20:20
Nitric acid can be formed in two steps from the atmospheric gases nitrogen and oxygen, plus hydrogen prepared by reforming natural gas. in the first step, nitrogen and hydrogen react to form ammonia: (g) (g) (g) in the second step, ammonia and oxygen react to form nitric acid and water: (g) (g) (g) (g) calculate the net change in enthalpy for the formation of one mole of nitric acid from nitrogen, hydrogen and oxygen from these reactions. round your answer to the nearest .
Answers: 3
You know the right answer?
7.0 g of nitrogen is reacted with 5.0 g of hydrogen to produce ammonia according to the chemical equ...
Questions

Mathematics, 12.07.2019 19:30




Mathematics, 12.07.2019 19:30

Computers and Technology, 12.07.2019 19:30



Mathematics, 12.07.2019 19:30

History, 12.07.2019 19:30



History, 12.07.2019 19:30



Mathematics, 12.07.2019 19:30

Mathematics, 12.07.2019 19:30