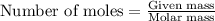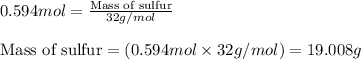Chemistry, 26.02.2020 04:18 welathyasf
Problem PageQuestion Sulfuric acid is essential to dozens of important industries from steelmaking to plastics and pharmaceuticals. More sulfuric acid is made than any other industrial chemical, and world production exceeds per year. The first step in the synthesis of sulfuric acid is usually burning solid sulfur to make sulfur dioxide gas. Suppose an engineer studying this reaction introduces of solid sulfur and of oxygen gas at into an evacuated tank. The engineer believes for the reaction at this temperature. Calculate the mass of solid sulfur he expects to be consumed when the reaction reaches equilibrium. Round your answer to significant digits. Note for advanced students: the engineer may be mistaken in his belief about the value of , and the consumption of sulfur you calculate may not be what he actually observes.

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 21:30
The density of an unknown gas at 98°c and 740 mmhg is 2.50 g/l. what is the molar mass of the gas with work showed?
Answers: 1

Chemistry, 22.06.2019 20:30
A40 kilogram skier starts at the top of a 12 meter high slope. at the bottom, she is travelling 10 meters per second. how much energy does she lose to friction
Answers: 2

Chemistry, 22.06.2019 23:00
What extra step distinguishes fermentation from glycolysis
Answers: 1

Chemistry, 23.06.2019 04:00
Which method would be best to separate a mixture of sand and gravel
Answers: 1
You know the right answer?
Problem PageQuestion Sulfuric acid is essential to dozens of important industries from steelmaking t...
Questions

English, 20.09.2019 18:30


Biology, 20.09.2019 18:30

Mathematics, 20.09.2019 18:30

History, 20.09.2019 18:30

Mathematics, 20.09.2019 18:30

Biology, 20.09.2019 18:30




Mathematics, 20.09.2019 18:30






Mathematics, 20.09.2019 18:30

History, 20.09.2019 18:30

Biology, 20.09.2019 18:30

Mathematics, 20.09.2019 18:30

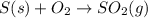
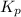 for above equation follows:
for above equation follows: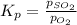
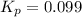
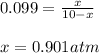
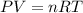
![650^oC=[650+273]K=923K](/tpl/images/0524/5297/e986f.png)
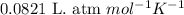

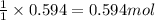 of sulfur
of sulfur