
Chemistry, 26.02.2020 01:51 kerarucker12pe384k
If an equal number of moles of reactants are used, do the following equilibrium mixtures contain primarily reactants or products? Please note this question has 1 submission for each part. (a) HCN(aq) + H2O(l) equilibrium reaction arrow CN −(aq) + H3O+(aq) K = 6.2 10-10 products reactants (b) H2(g) + Cl2(g) equilibrium reaction arrow 2 HCl(g) K = 2.51 104 reactants products

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 13:20
Determine which intermolecular forces are the dominant (strongest) forces for a pure sample of each of the following molecules by placing the molecules into the correct bins. drag the appropriate molecular formula to their respective bins.
Answers: 3

Chemistry, 21.06.2019 22:00
Match each object to its description: a. coma of a comet b. comet tail c. oort cloud haze surrounding a nucleus created by solar wind. hypothetical sphere around the solar system
Answers: 1

Chemistry, 22.06.2019 14:30
How can carbon move from "land" to bodies of water? describe the way human impact has lead to increased levels of co2 in the atmosphere.
Answers: 2

Chemistry, 22.06.2019 23:30
The comparison of the number of atoms in a copper coin the size of a penny with the number of people on earth is made to illustrate which of the following? a. that atoms are indivisible b. that atoms are very small c. that atoms are very large d. that in a copper penny, there is one atom for every person on earth
Answers: 1
You know the right answer?
If an equal number of moles of reactants are used, do the following equilibrium mixtures contain pri...
Questions

Social Studies, 29.09.2019 13:00

History, 29.09.2019 13:00

Biology, 29.09.2019 13:00




English, 29.09.2019 13:00

History, 29.09.2019 13:00


Chemistry, 29.09.2019 13:00




Physics, 29.09.2019 13:00

Physics, 29.09.2019 13:00


Computers and Technology, 29.09.2019 13:00

Mathematics, 29.09.2019 13:00

History, 29.09.2019 13:00

History, 29.09.2019 13:00

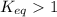 ; the reaction is product favored.When
; the reaction is product favored.When 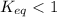 ; the reaction is reactant favored.When
; the reaction is reactant favored.When 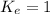 ; the reaction is in equilibrium.
; the reaction is in equilibrium.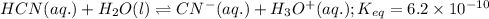
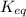 for above reaction follows:
for above reaction follows:![K_{eq}=\frac{[CN^-][H_3O^+]}{[HCN][H_2O]}=6.2\times 10^{-10}](/tpl/images/0524/2318/abbf6.png)
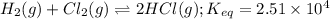
![K_{eq}=\frac{[HCl]^2}{[H_2][Cl_2]}=2.51\times 10^{4}](/tpl/images/0524/2318/4af57.png)


