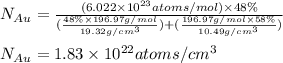Gold forms a substitutional solid solution with silver. Compute the number of gold atoms per cubic centimeter for a silver-gold alloy that contains 42 wt% Au and 58 wt% Ag. The densities of pure gold and silver are 19.32 and 10.49 g/cm3, respectively. The atomic weight of Au is 196.97 g/mol.

Answers: 1


Another question on Chemistry

Chemistry, 21.06.2019 19:30
If the element whose electric configuration ends in the d sublevel, the element is calssified as? a.inner transition b.noble gases c.representative d. transition
Answers: 2

Chemistry, 22.06.2019 08:00
Identify a strong intermolecular force of attraction between an alcohol
Answers: 1

Chemistry, 23.06.2019 00:30
How can you write e method for the experiment of separating sand from water by filtration process? 1-materials 2-steps 3-conclusion also the same for the separating process of water and salt by filtration or distillation. quick because i need to finish my hw
Answers: 2

Chemistry, 23.06.2019 01:00
Wind and moving water provide energy. chemical mechanical thermal none of the above
Answers: 1
You know the right answer?
Gold forms a substitutional solid solution with silver. Compute the number of gold atoms per cubic c...
Questions

Social Studies, 17.10.2019 14:00

Mathematics, 17.10.2019 14:00




Chemistry, 17.10.2019 14:00



Chemistry, 17.10.2019 14:00


History, 17.10.2019 14:00

Business, 17.10.2019 14:00

Mathematics, 17.10.2019 14:00




Mathematics, 17.10.2019 14:00




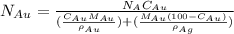
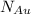 = number of gold atoms per cubic centimeters
= number of gold atoms per cubic centimeters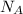 = Avogadro's number =
= Avogadro's number = 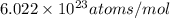
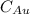 = Mass percent of gold in the alloy = 42 %
= Mass percent of gold in the alloy = 42 %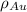 = Density of pure gold =
= Density of pure gold = 
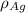 = Density of pure silver =
= Density of pure silver = 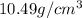
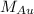 = molar mass of gold = 196.97 g/mol
= molar mass of gold = 196.97 g/mol