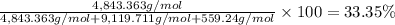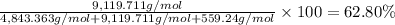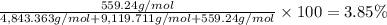Chemistry, 25.02.2020 03:52 TravKeepIt100
Convert the composition of the following alloy from atom percent to weight percent (a) 44.9 at% of silver, (b) 46.3 at% of gold, and (c) 8.8 at% of copper. The atomic weights for silver, gold, and copper are 107.87, 196.97, and 63.55 g/mol, respectively.

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 06:40
Three alkali metals in group 1 are a. calcium, strontium, barium b. boron, aluminum, gallium c. sodium, potassium, rubidium d. fluorine, iodine, chlorine
Answers: 1


Chemistry, 22.06.2019 22:30
You just calculated that the heat of fusion for chloromethane is 6400 j/mol. the heat of fusion for hydrogen is 120 j/mol.? which of the following account for this difference? more than one correcta. chloromethane can absorb more energy at the same temperature. b. hydrogen has stronger intermolecular forces than chloromethane. c. hydrogen molecules can pack more closely than chloromethane molecules. d. chloromethane experiences dipole-dipole interactions. e. chloromethane has a higher molar mass than hydrogen.
Answers: 3

Chemistry, 23.06.2019 03:00
What volume does 1.70 ×10–3 mol of chlorine gas occupy if its temperature is 20.2 °c and its pressure is 795 mm hg?
Answers: 3
You know the right answer?
Convert the composition of the following alloy from atom percent to weight percent (a) 44.9 at% of s...
Questions


Mathematics, 02.09.2019 02:00

Chemistry, 02.09.2019 02:00

History, 02.09.2019 02:00


Social Studies, 02.09.2019 02:00


English, 02.09.2019 02:00






English, 02.09.2019 02:00


Mathematics, 02.09.2019 02:00



History, 02.09.2019 02:00

Mathematics, 02.09.2019 02:00

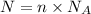
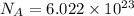 = Avogadro number
= Avogadro number