
Chemistry, 24.02.2020 18:53 jaylennkatrina929
A certain second-order reaction (B→products) has a rate constant of 1.30×10−3 M−1⋅s−1 at 27 ∘C and an initial half-life of 224 s . What is the concentration of the reactant B after one half-life?

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 18:00
Alidded glass container is filled with a colored gas. after a period of time, it is observed that the gas is uniformly spread throughout the box and that the movement has slowed considerably. next, a warm iron plate is carefully placed under the box. why is there resumed movement of the gas in the container?
Answers: 2

Chemistry, 22.06.2019 18:30
You open a can of soda at room temperature and hear a hiss. which of the following factors has changed inside the container? a.) atmospheric pressure b.) temperature of gas c.) type of gas d.) amount of gas
Answers: 1

Chemistry, 22.06.2019 19:10
How does the atmosphere to make earth livable? check all that apply. causes the seasons contains oxygen provides warmth creates important nutrients blocks harmful energy from the sun plz like !
Answers: 2

Chemistry, 22.06.2019 19:30
To calculate percent by mass, use the equation below: calculate the percent by mass of each element. %n = % %h = % %o = %
Answers: 3
You know the right answer?
A certain second-order reaction (B→products) has a rate constant of 1.30×10−3 M−1⋅s−1 at 27 ∘C and a...
Questions

Mathematics, 02.07.2021 07:30


Mathematics, 02.07.2021 07:30

Mathematics, 02.07.2021 07:30

Mathematics, 02.07.2021 07:30


Health, 02.07.2021 07:30

Mathematics, 02.07.2021 07:30



Mathematics, 02.07.2021 07:30



Physics, 02.07.2021 07:40

English, 02.07.2021 07:40

Mathematics, 02.07.2021 07:40

Mathematics, 02.07.2021 07:40

English, 02.07.2021 07:40

Advanced Placement (AP), 02.07.2021 07:40


![\dfrac{d[B]}{dt}=-k[B]^2](/tpl/images/0521/4312/89d63.png)
![\dfrac{1}{[B]}=\dfrac{1}{[B]_0}+kt](/tpl/images/0521/4312/c6bcf.png)
![t_{1/2}=\dfrac{1}{k[A]_0}](/tpl/images/0521/4312/90d03.png)
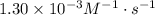 and the half-life time 224s to find [A]₀:
and the half-life time 224s to find [A]₀:![224s=\dfrac{1}{1.30\times10^{-3}M^{-1}\cdot s^{-1}[A]_0}](/tpl/images/0521/4312/bcef9.png)
![[A]_o=0.291M](/tpl/images/0521/4312/294dc.png)


