Given the equation representing a reaction: H + H ---> H₂What occurs during this reaction? a. A bond is broken and energy is absorbed. b. A bond is broken and energy is released. c. A bond is formed and energy is absorbed. d. A bond is formed and energy is released.

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 22:30
200. ml of 3.00 m nacl solution is diluted to a final volume of 500. ml. what is the molarity of the final solution?
Answers: 2

Chemistry, 22.06.2019 07:10
Remember to use the proper number of significant figures and leading zeros in all calculations.gelatin has a density of 1.27 g/cm³. if you have a blob of gelatin dessert that fills a 2.0 liter bottle, what is its mass? 2540 g2500 g3.9 x 10-43.937x 10-4
Answers: 3

Chemistry, 22.06.2019 10:50
8) a mixture of he, ne and ar has a pressure of 7.85 atm. if the ne has a mole fraction of 0.47 and 8) ar has a mole fraction of 0.23, what is the pressure of he? a) 4.2 atm b) 3.7 atm c) 5.5 atm d) 2.4 atm e) 1.8 atm
Answers: 1

Chemistry, 22.06.2019 22:30
You just calculated that the heat of fusion for chloromethane is 6400 j/mol. the heat of fusion for hydrogen is 120 j/mol.? which of the following account for this difference? more than one correcta. chloromethane can absorb more energy at the same temperature. b. hydrogen has stronger intermolecular forces than chloromethane. c. hydrogen molecules can pack more closely than chloromethane molecules. d. chloromethane experiences dipole-dipole interactions. e. chloromethane has a higher molar mass than hydrogen.
Answers: 3
You know the right answer?
Given the equation representing a reaction: H + H ---> H₂What occurs during this reaction? a. A b...
Questions





English, 29.06.2019 15:00


Mathematics, 29.06.2019 15:00




Mathematics, 29.06.2019 15:00


Mathematics, 29.06.2019 15:00



Biology, 29.06.2019 15:00

Biology, 29.06.2019 15:00

Chemistry, 29.06.2019 15:00


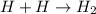 (ΔH = Positive)
(ΔH = Positive)