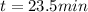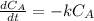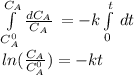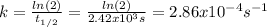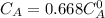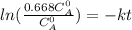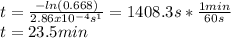Answers: 1


Another question on Chemistry


Chemistry, 22.06.2019 10:30
Asample of air with a volume of 2.20m3 at a pressure of 105 kpa and a temperature of 30c is cooled to 10c and the pressure is reduced to 75.0 kpa. what is the new volume? 6.9 1.34 2.56 43.0 2.88
Answers: 1

Chemistry, 22.06.2019 12:00
Ican determine the molar mass of an element by looking on the under the atomic mass for the element. for example the molar mass of phosphorus is 30.974 grams/mole. avogadro’s number tells me the amount of representative particles in 1 mole of any substance. this means 12.011 gram sample of carbon and a 32.0 gram sample of sulfur have the same number of atoms.
Answers: 1

Chemistry, 22.06.2019 19:30
Estimate the molar mass of the gas that effuses at 1.6 times the effusion rate of carbon dioxide.
Answers: 1
You know the right answer?
The decomposition reaction of A to B is a first-order reaction with a half-life of 2.42×103 seconds:...
Questions

History, 21.12.2021 07:00

English, 21.12.2021 07:00











English, 21.12.2021 07:10


Mathematics, 21.12.2021 07:10


Health, 21.12.2021 07:10

Mathematics, 21.12.2021 07:10

Mathematics, 21.12.2021 07:10