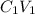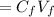Chemistry, 20.02.2020 08:17 Wolfgirl2032
A biochemist carefully measures the molarity of magnesium ion in 47, mL of cell growth medium to be 97. ??. Unfortunately, a careless graduate student forgets to cover the container of growth medium and a substantial amount of the solvent evaporates. The volume of the cell growth medium falls to 6.0 mL. Calculate the new molarity of magnesium ion in the cell growth medium Be sure your answer has the correct number of significant digits. HM x 10

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 19:00
How does a catalyst increase the speed of a reaction? a. the catalyst eliminates the activated complex stage, allowing products to form immediately. b. the catalyst lowers the energy level of the reactants, making it easier for them to react. c. the catalyst makes it easier for the activated complex to form, lowering the activation energy. d. the catalyst raises the energy level of the products, making the reaction finish sooner. reset next
Answers: 1

Chemistry, 23.06.2019 01:00
Which substance—wood or silver—is the better thermal conductor? a thermal conductor is a material that requires very little heat energy to change its temperature. explain your answer.
Answers: 3

Chemistry, 23.06.2019 02:00
What are fossils of organisms that existed over a wide area but only for a limited time period called?
Answers: 2

Chemistry, 23.06.2019 09:30
Sheela and her brother hari were sitting in the living room, watching tv. suddenly hari said that he thinks something is burning in the other room. how did he get the burning smell?
Answers: 3
You know the right answer?
A biochemist carefully measures the molarity of magnesium ion in 47, mL of cell growth medium to be...
Questions



Mathematics, 31.03.2020 22:45


Health, 31.03.2020 22:45






Mathematics, 31.03.2020 22:45




Mathematics, 31.03.2020 22:45

English, 31.03.2020 22:45

Social Studies, 31.03.2020 22:46

Mathematics, 31.03.2020 22:46

Biology, 31.03.2020 22:46

Mathematics, 31.03.2020 22:46