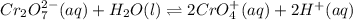Chemistry, 18.02.2020 21:48 Kennedy3449
Consider an acidic chromate-dichromate system at equilibrium in which the color is an orange-yellow. The reaction is represented by the equation:Cr2O72-(aq) + H2O(l)↔ CrO42+(aq) + H+(aq) orange yellow Which of the following best describes what happens if a strong base is added to the system at equilibrium? Explain your answer.

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 00:30
Jessica is traveling from miami, florida, to chicago, illinois. using the map, tell one way the land will change during the second half of her trip.
Answers: 1

Chemistry, 22.06.2019 09:00
This chart lists four kinds of polymers and their sources. what can be known about all four polymers, despite their differences? they come from living things. they share ionic carbon bonds. they are at least 100 monomers long. they are made of repeating subunits.
Answers: 2

Chemistry, 22.06.2019 11:10
Which of the following shapes would represent a molecule with two bonded atoms and 3 lone pairs on only one of them , trigonal planar , bent , trigonal pyramidal , linear
Answers: 1

Chemistry, 22.06.2019 13:40
Can someone me with 6 to 10 plz this is for masteries test.
Answers: 1
You know the right answer?
Consider an acidic chromate-dichromate system at equilibrium in which the color is an orange-yellow....
Questions


Mathematics, 03.07.2019 07:00

Physics, 03.07.2019 07:00


Mathematics, 03.07.2019 07:00


Mathematics, 03.07.2019 07:00


Chemistry, 03.07.2019 07:00




Arts, 03.07.2019 07:00

Biology, 03.07.2019 07:00



Mathematics, 03.07.2019 07:00

Biology, 03.07.2019 07:00

Arts, 03.07.2019 07:00