
Chemistry, 18.02.2020 21:22 leandrogarin37p2g5ds
Phosphoric acid is a triprotic acid ( K a1 = 6.9 × 10 − 3 Ka1=6.9×10−3, K a2 = 6.2 × 10 − 8 Ka2=6.2×10−8, and K a3 = 4.8 × 10 − 13 Ka3=4.8×10−13). To find the pH of a buffer composed of H 2 PO − 4 ( aq ) H2PO4−(aq) and HPO 2 − 4 ( aq ) HPO42−(aq) , which p K a Ka value should be used in the Henderson–Hasselbalch equation?

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 09:50
Although respiratory organs vary across different organisms, they all contain respiratory surfaces that have a large surface area and are extremely thin. explain why having an extremely thin respiratory surface with a large surface area is advantageous for the process of gas exchange
Answers: 1

Chemistry, 22.06.2019 14:30
Select the word from the list that best fits the definition the nuclear family into which a person is born or adopted.
Answers: 2

Chemistry, 22.06.2019 16:50
Assuming complete dissociation of the solute, how many grams of kno3 must be added to 275 ml of water to produce a solution that freezes at -14.5 c? the freezing point for pure water is 0.0 c and k_f is equal to 1.86 c/m
Answers: 3

Chemistry, 23.06.2019 00:30
Element j is 1s 2s 2p 3s . (i) how many unpaired electrons does j have? (ii) is j a good oxidizing agent or a reducing agent? (iii) state reason for the answer.
Answers: 1
You know the right answer?
Phosphoric acid is a triprotic acid ( K a1 = 6.9 × 10 − 3 Ka1=6.9×10−3, K a2 = 6.2 × 10 − 8 Ka2=6.2×...
Questions

Mathematics, 15.06.2021 17:00

Mathematics, 15.06.2021 17:00


Social Studies, 15.06.2021 17:00


Spanish, 15.06.2021 17:00



Mathematics, 15.06.2021 17:00



Mathematics, 15.06.2021 17:00



English, 15.06.2021 17:00

Mathematics, 15.06.2021 17:00

Mathematics, 15.06.2021 17:00

Biology, 15.06.2021 17:00

Mathematics, 15.06.2021 17:00

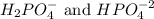 , we use the
, we use the 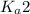
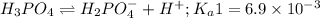
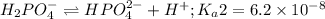
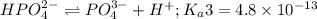
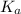 of second dissociation process
of second dissociation process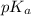 , we use the equation:
, we use the equation: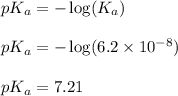
![pH=pK_a2+\log(\frac{[\text{conjugate base}]}{[\text{weak acid}]})](/tpl/images/0514/6294/e7d91.png)
![pH=pK_a2+\log(\frac{[HPO_4^{2-}]}{[H_2PO_4^-]})](/tpl/images/0514/6294/f63a6.png)
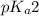 = negative logarithm of second acid dissociation constant of phosphoric acid = 7.21
= negative logarithm of second acid dissociation constant of phosphoric acid = 7.21![[HPO_4^{2-}]](/tpl/images/0514/6294/c0ca9.png) = concentration of conjugate base
= concentration of conjugate base![[H_2PO_4^{-}]](/tpl/images/0514/6294/fcc52.png) = concentration of weak acid
= concentration of weak acid

