
Chemistry, 18.02.2020 03:29 onewaydemon
The iodide ion reacts with hypochlorite ion (the active ingredient in chlorine bleaches) in the following way:
OCl- + I- → OI-1 +Cl-.
This rapid reaction gives the following rate data:
[OCl-](M) [I]- (M) Rate (M/s)
1.5×10^3 1.5×10^3 1.36×10^4
3.0×10^3 1.5×10^3 2.72×10^4
1.5×10^3 3.0×10^3 2.72×10^4
Write the rate law for this reaction.
Calculate the rate constant with proper units.
Calculate the rate when [OCl-]= 1.8×10^3 M and [I-]= 6.0×10^4 M .

Answers: 2


Another question on Chemistry


Chemistry, 22.06.2019 18:00
Hydrogenation reactions, in which h2 and an "unsaturated" organic compound combine, are used in the food, fuel, and polymer industries. in the simplest case, ethene (c2h4) and h2 form ethane (c2h6). if 140 kj is given off per mole of c2h4 reacting, how much heat (in mj) is released when 12 kg of c2h6 forms?
Answers: 2

Chemistry, 22.06.2019 21:00
What type of radiation is lead emitting in the following equation? alpha particles beta particles gamma rays
Answers: 3

Chemistry, 22.06.2019 21:20
Phosgene (carbonyl chloride), cocl2, is an extremely toxic gas that is used in manufacturing certain dyes and plastics. phosgene can be produced by reacting carbon monoxide and chlorine gas at high temperatures: co(g) cl2(g)⇌cocl2(g) carbon monoxide and chlorine gas are allowed to react in a sealed vessel at 477 ∘c . at equilibrium, the concentrations were measured and the following results obtained: gas partial pressure (atm) co 0.830 cl2 1.30 cocl2 0.220 what is the equilibrium constant, kp, of this reaction
Answers: 2
You know the right answer?
The iodide ion reacts with hypochlorite ion (the active ingredient in chlorine bleaches) in the foll...
Questions

Mathematics, 31.10.2020 21:10

Mathematics, 31.10.2020 21:10

Mathematics, 31.10.2020 21:10

Mathematics, 31.10.2020 21:10

Mathematics, 31.10.2020 21:10

Mathematics, 31.10.2020 21:10

Arts, 31.10.2020 21:10


Mathematics, 31.10.2020 21:10

Mathematics, 31.10.2020 21:10

Chemistry, 31.10.2020 21:10

English, 31.10.2020 21:10

Mathematics, 31.10.2020 21:10

Chemistry, 31.10.2020 21:10



Physics, 31.10.2020 21:10

English, 31.10.2020 21:10

History, 31.10.2020 21:10

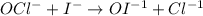
![K \times [OCl^{-}] \times [l^{-}]](/tpl/images/0513/8261/88013.png)
![K [OCl^{-}][l^{-}]](/tpl/images/0513/8261/94fd7.png)
![[OCl^{-}] = [l^{-}]](/tpl/images/0513/8261/5e8f1.png) )
)

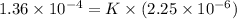
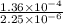
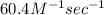

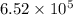
![[OCl^{-}] = 1.8 \times 10^{3}](/tpl/images/0513/8261/c42db.png) M and
M and ![[I^{-}]= 6.0 \times 10^{4}](/tpl/images/0513/8261/40481.png) M is
M is 

