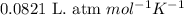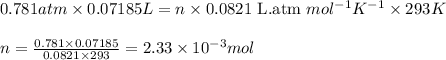Chemistry, 18.02.2020 01:59 MariaGuerra
You analyze a sample of unknown metal as you would in this experiment. You measure the volume of H2(g) generated to be 71.85 mL and the water temperature to be 20.0°C. You calculate PH2 to be 0.781 atm. Use the ideal gas law to calculate the number of moles of hydrogen gas generated.

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 17:30
A650 ml sodium bromine solution has a bromide ion concentration of 0.245 m. what is the mass (g) of sodium bromide in solution? a) 103.b)0.00155.c)16400.d) 16.4.e) 0.159
Answers: 2

Chemistry, 22.06.2019 23:00
What is the number of neutrons in an atom with atomic mass of 35
Answers: 2

Chemistry, 23.06.2019 00:30
There are approximately 15 milliliters (ml) in 1 tablespoon (tbsp). what expression can be used to find the approximate number of milliliters in 3 tbsp?
Answers: 1

Chemistry, 23.06.2019 01:20
Use the de broglie's wave equation to find the wavelength of an electron moving at 4.5 × 106 m/s. show your work. note: h= plank's constant (6.62607 x 10-34 j s)
Answers: 1
You know the right answer?
You analyze a sample of unknown metal as you would in this experiment. You measure the volume of H2(...
Questions

Mathematics, 09.09.2021 05:50

Biology, 09.09.2021 05:50

Mathematics, 09.09.2021 05:50






Mathematics, 09.09.2021 05:50


Mathematics, 09.09.2021 05:50



Chemistry, 09.09.2021 05:50

Mathematics, 09.09.2021 05:50

Mathematics, 09.09.2021 05:50


Mathematics, 09.09.2021 05:50

Mathematics, 09.09.2021 05:50

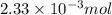
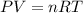
![20^oC=[20+273]K=293K](/tpl/images/0513/7044/3b5d4.png)