
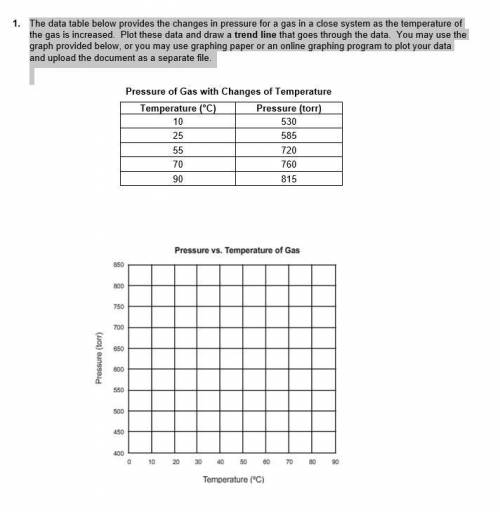
1. The data table below provides the changes in pressure for a gas in a close system as the temperature of the gas is increased. Plot these data and draw a trend line that goes through the data. You may use the graph provided below, or you may use graphing paper or an online graphing program to plot your data and upload the document as a separate file.
• Calculate the slope of the trend line including the units. Be sure to show all steps of your calculations.
• How are the variables of temperature and pressure related to one another?
• Use the trend line to determine what will be the gas pressure at 0°C.
i have attached a picture that shows the data tables

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 18:00
Acylinder is filled with 2.00 moles of nitrogen, 3.00 moles of argon and 5.00 moles of helium. if the gas mixture is at stp, what is the partial pressure of the argon
Answers: 1

Chemistry, 22.06.2019 00:30
Which best describes why nh4+ can form an ionic bond with ci-?
Answers: 1

Chemistry, 22.06.2019 06:00
An atom of lithium (li) and an atom of chlorine (cl) engage in a chemical reaction. which correctly describes the structure of the resulting chemical compound? hint: consider the class of each element. the chemical compound will have a network structure. the chemical compound will have triple bonds. the chemical compound will have a ball-and-stick structure. the chemical compound will have double bonds.
Answers: 2

Chemistry, 22.06.2019 12:20
The yearly amounts of carbon emissions from cars in belgium are normally distributed with a mean of 13.9 gigagrams per year and a standard deviation of 5.8 gigagrams per year. find the probability that the amount of carbon emissions from cars in belgium for a randomly selected year are between 11.5 gigagrams and 14.0 gigagrams per year. a. 0.340 b. 0.660 c. 0.167 d. 0.397
Answers: 2
You know the right answer?
1. The data table below provides the changes in pressure for a gas in a close system as the temperat...
Questions

English, 15.03.2020 02:58



Mathematics, 15.03.2020 02:58

Mathematics, 15.03.2020 02:59


French, 15.03.2020 02:59

Chemistry, 15.03.2020 02:59

History, 15.03.2020 02:59


Mathematics, 15.03.2020 02:59


English, 15.03.2020 03:00


History, 15.03.2020 03:01



History, 15.03.2020 03:01


Mathematics, 15.03.2020 03:01



