Chemistry, 16.02.2020 19:41 queenkimm26
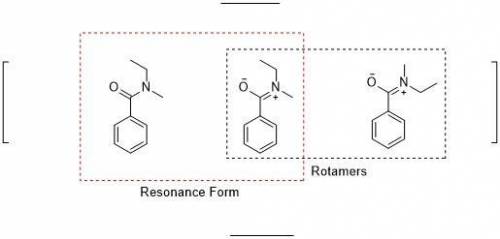
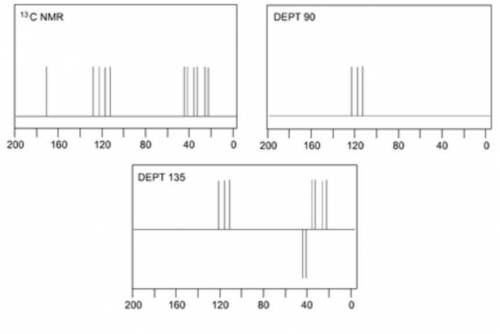
When an unknown amine reacts with an unknown acid chloride, an amide with a molecular mass of 163 g/mol (M = 163 m/z) is formed. In the infrared spectrum, important absorptions appear at 1661, 750 and 690 cm–1. The 13C NMR and DEPT spectra are provided below. Draw the structure of the product as the resonance contributor lacking any formal charges.

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 01:50
7. what temperature is need to just dissolve 50 g of nh4cl in 75 g of water? '
Answers: 1



Chemistry, 22.06.2019 21:30
How many liters of 3.0 m naoh solution will react with 0.60 liters of 4.0 m h2so4? h2so4 + naoh → na2so4 + h2o 1.2 l 1.6 l 2.4 l 2.8 l
Answers: 3
You know the right answer?
When an unknown amine reacts with an unknown acid chloride, an amide with a molecular mass of 163 g/...
Questions




Biology, 15.04.2020 15:34





Chemistry, 15.04.2020 15:34









Mathematics, 15.04.2020 15:34


Biology, 15.04.2020 15:34