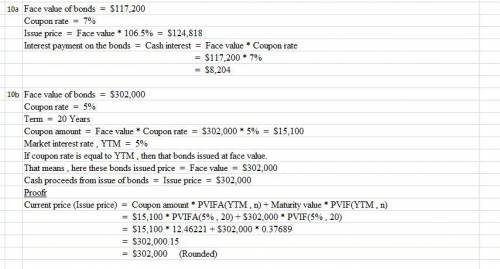
Ava, Inc., issued 7% bonds, dated January 1, with a face amount of $117,200 on January 1, 2016 for an issue price of 106.5. The bonds mature on December 31, 2025 (10 years). For bonds of similar risk and maturity the market yield is 9%. Interest is paid annually on December 31.
What is the interest payment on the bond?
10b) A company issued 5%, 20-year bonds with a face amount of $302,000. The market yield for bonds of similar risk and maturity is 5%. Interest is paid annually. What is the debit to cash for the bond proceeds?
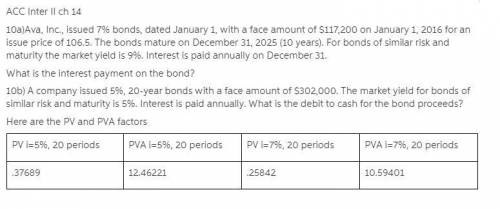
Here are the PV and PVA factors
PV i=5%, 20 periods
PVA i=5%, 20 periods
PV i=7%, 20 periods
PVA i=7%, 20 periods
.37689
12.46221
.25842
10.59401

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 22:00
Fission of uranium-235 products energy and a. isotopes of smaller elements b. isotopes of larger elements c. lighter isotopes of uranium d. heavier isotopes of uranium
Answers: 3

Chemistry, 22.06.2019 12:00
Ican determine the molar mass of an element by looking on the under the atomic mass for the element. for example the molar mass of phosphorus is 30.974 grams/mole. avogadro’s number tells me the amount of representative particles in 1 mole of any substance. this means 12.011 gram sample of carbon and a 32.0 gram sample of sulfur have the same number of atoms.
Answers: 1

Chemistry, 23.06.2019 04:31
What are the coefficients that will balance the skeleton equation below? n2 + h2 → nh3
Answers: 1

Chemistry, 23.06.2019 15:30
How many grams of c3h8 is needed in the reactants to produce 10.5 mil of h2o
Answers: 2
You know the right answer?
Ava, Inc., issued 7% bonds, dated January 1, with a face amount of $117,200 on January 1, 2016 for a...
Questions

Business, 28.04.2021 20:00

Computers and Technology, 28.04.2021 20:00




Mathematics, 28.04.2021 20:00


Social Studies, 28.04.2021 20:00

Mathematics, 28.04.2021 20:00

English, 28.04.2021 20:00

Mathematics, 28.04.2021 20:00