
You double the partial pressure of a gas over a liquid at constant temperature. Which of these statements is then true? a. The Henry’s Law constant is decreased by half. b. There are half as many gas molecules in the liquid. c. There is no change in the number of gas molecules in the liquid. d. The Henry’s Law constant is doubled. e. There are twice as many gas molecules in the liquid.

Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 17:10
Abullet found at a crime scene may be used as evidence in a trial if the percentage of metals match to the composition of metals in a bullet from the suspect's ammunition. a forensic scientist's analysis of the bullet shows that it contains 11.9 g of lead, 0.5 g of tin, and 0.8 b of antimony. what is the percentage of lead metal in the bullet? express your answers to the one's place.
Answers: 2

Chemistry, 22.06.2019 09:00
Chemical energy is a form of a. kinetic energy only. b. both potential and kinetic energy. c. neither potential nor kinetic energy. d. potential energy only. reset
Answers: 1

Chemistry, 22.06.2019 10:00
According to the tide table below what time of day will the highest tide occur? (2 pt) the highest tide will occur at
Answers: 1

Chemistry, 22.06.2019 10:30
Acompound has a molar mass of 92.02 grams/mole, and its percent composition is 30.4% nitrogen (n) and 69.6% oxygen (o). what is its molecular formula? a. n2o4 b. no2 c. n2o d. n4o2
Answers: 1
You know the right answer?
You double the partial pressure of a gas over a liquid at constant temperature. Which of these state...
Questions

History, 19.10.2019 21:30

Mathematics, 19.10.2019 21:30

Mathematics, 19.10.2019 21:30



Health, 19.10.2019 21:30


English, 19.10.2019 21:30

Mathematics, 19.10.2019 21:30

History, 19.10.2019 21:30


Arts, 19.10.2019 21:30



Mathematics, 19.10.2019 21:30

History, 19.10.2019 21:50


English, 19.10.2019 21:50


Mathematics, 19.10.2019 21:50

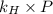
 = Henry's constant
= Henry's constant

