Chemistry, 14.02.2020 02:48 seiglersteven99
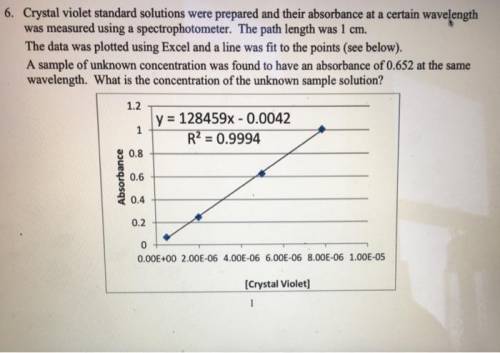
Crystal violet standard solutions were prepared and their absorbance at a certain wavelength was measured using a spectrophotometer. The path length was 1 cm. The data was plotted using Excel and a line was fit to the points (see below). A sample of unknown concentration was found to have an absorbance of 0.652 at the same wavelength. (a) What is the molar absorptivity of this compound at the certain wavelength? (b) What is the concentration of the unknown sample solution?

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 13:30
One of the reactions in a blast furnace used to reduce iron is shown above. how many grams of fe2o3 are required to produce 15.5 g of fe if the reaction occurs in the presence of excess co? a.11.1 g b.22.1 g c.30.0 g d.44.2 g
Answers: 2

Chemistry, 22.06.2019 04:00
Tin has ten stable isotopes. the heaviest, 124sn, makes up 5.80% of naturally occuring tin atoms. how many atoms of 124sn are present in 82.0 g of naturally occurring tin? what is the total mass of the 124sn atoms in this sample?
Answers: 3


Chemistry, 22.06.2019 14:30
Aroom with dimensions 7.00m×8.00m×2.50m is to be filled with pure oxygen at 22.0∘c and 1.00 atm. the molar mass of oxygen is 32.0 g/mol. how many moles noxygen of oxygen are required to fill the room? what is the mass moxygen of this oxygen?
Answers: 1
You know the right answer?
Crystal violet standard solutions were prepared and their absorbance at a certain wavelength was mea...
Questions



Biology, 20.11.2019 18:31

Social Studies, 20.11.2019 18:31

History, 20.11.2019 18:31

Mathematics, 20.11.2019 18:31

Health, 20.11.2019 18:31

Mathematics, 20.11.2019 18:31



Mathematics, 20.11.2019 18:31



Mathematics, 20.11.2019 18:31


History, 20.11.2019 18:31




English, 20.11.2019 18:31