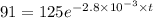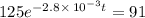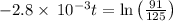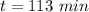The decomposition of dinitrogen pentoxide, N2O5, to NO2 and O2 is a first-order reaction. At 60°C, the rate constant is 2.8 × 10-3min-1. If a rigid vessel initially contains only N2O5 at a pressure of 125 kPa, how long will it take for the total pressure to reach 176 kPa?
a. 113 min
b. 129 min
c. 42 min
d. 182 min
e. 62 min
f. 83 min

Answers: 3


Another question on Chemistry

Chemistry, 22.06.2019 02:10
When 225mg of anthracene, c14h10(s), was burned in a bomb calorimeter the temperature rose by 1.75k. calculate the calorimeter constant. by how much will the temperature rise when 125mg of phenol, c6h5oh(s), is burned in the calorimeter under the same conditions? (δch< (c14h10,s)=–7061 kj mol−1.)
Answers: 3

Chemistry, 22.06.2019 16:00
Which of the following is the correct definition of chemical energy? a. energy an object has because of its motion or position b. energy resulting from the flow of charged particles, such as electrons or ions c. energy produced from the splitting of atoms d. energy stored in chemical bonds of molecules
Answers: 1

Chemistry, 22.06.2019 17:30
What will most likely happen in the absence of a cell membrane? a) photosynthesis will not take place. b) the cell will not store food, water, nutrients, and waste. c) energy will not be released during cellular respiration. d) substances will pass in and out of the cell in an uncontrolled manner.
Answers: 1

Chemistry, 23.06.2019 01:00
How does carbon monoxide pose the greatest threat to humans? a. it can be produced by wood fires. b. it can be produced by home furnaces. c. it is produced by acid rain. d. it is produced by modern automobiles.
Answers: 2
You know the right answer?
The decomposition of dinitrogen pentoxide, N2O5, to NO2 and O2 is a first-order reaction. At 60°C, t...
Questions




Mathematics, 01.02.2022 23:50

Spanish, 01.02.2022 23:50

Social Studies, 01.02.2022 23:50

Computers and Technology, 01.02.2022 23:50



Mathematics, 01.02.2022 23:50


Mathematics, 01.02.2022 23:50

English, 01.02.2022 23:50

Spanish, 01.02.2022 23:50


Mathematics, 01.02.2022 23:50


Chemistry, 01.02.2022 23:50

Computers and Technology, 01.02.2022 23:50

Mathematics, 01.02.2022 23:50

![[A_t]=[A_0]e^{-kt}](/tpl/images/0510/6499/1ef89.png)
![[A_t]](/tpl/images/0510/6499/5262c.png) is the concentration at time t
is the concentration at time t
![[A_0]](/tpl/images/0510/6499/9a686.png) is the initial concentration
is the initial concentration
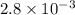 min⁻¹
min⁻¹