
Chemistry, 13.02.2020 20:57 mapoohdoll
Initially, a closed flask contains a mixture of N2 at a partial pressure of 3 atm and H2 at a partial pressure of 5 atm. The mixture reaches equilibrium and the partial pressure of NH3 is 2 atm. What is the value of the equilibrium constant?

Answers: 1


Another question on Chemistry

Chemistry, 22.06.2019 14:30
The valence of aluminum is +3, and the valence of the chlorine is -1. the formula fir the aluminum chloride is correctly written as
Answers: 2

Chemistry, 22.06.2019 19:30
Use the periodic table to find the molar mass of each element. molar mass h = g/mol molar mass s = g/mol molar mass o = g/mol
Answers: 3

Chemistry, 22.06.2019 21:00
Which answer tells the reason the earth’s climate is getting warmer? too many animals are becoming extinct. large glaciers are melting in antarctica. the earth is moving closer to the sun. driving cars gives off gases that trap heat in the atmosphere.
Answers: 1

Chemistry, 23.06.2019 00:20
How many lone pairs of electrons are on the central atom of no3- and what is the molecular shape? one, trigonal planar zero, trigonal pyramidal zero, trigonal planar one, tetrahedral one, trigonal pyramidal
Answers: 1
You know the right answer?
Initially, a closed flask contains a mixture of N2 at a partial pressure of 3 atm and H2 at a partia...
Questions


Mathematics, 12.01.2020 20:31


Biology, 12.01.2020 20:31

Chemistry, 12.01.2020 20:31


Chemistry, 12.01.2020 20:31

Social Studies, 12.01.2020 20:31




Mathematics, 12.01.2020 20:31

Physics, 12.01.2020 20:31

Mathematics, 12.01.2020 20:31





Mathematics, 12.01.2020 20:31

Mathematics, 12.01.2020 20:31

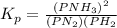 Hence we have
Hence we have

