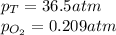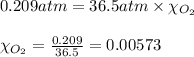Chemistry, 13.02.2020 05:14 jrassicworld4ever
What should be the mole fraction of O2 in the gas mixture the diver breathes in order to have the same partial pressure of oxygen in his lungs as he would at sea level? Note that the mole fraction of oxygen at sea level is 0.209.

Answers: 3


Another question on Chemistry

Chemistry, 21.06.2019 15:30
1. baking powder is a 1: 1 molar mixture of cream of tartar (khc4h4o6) and baking soda (nahco3). a recipe calls for two teaspoons (a total of 8.0 grams) of cream of tartar. how much baking soda must be added for both materials to react completely?
Answers: 2

Chemistry, 22.06.2019 10:30
Asample of air with a volume of 2.20m3 at a pressure of 105 kpa and a temperature of 30c is cooled to 10c and the pressure is reduced to 75.0 kpa. what is the new volume? 6.9 1.34 2.56 43.0 2.88
Answers: 1

Chemistry, 22.06.2019 18:10
The atom fluorine generally will become stable through the formation of an ionic chemical compound by accepting electron(s) from another atom. this process will fill its outer energy level of electrons.
Answers: 1

Chemistry, 23.06.2019 01:00
Chromium(iii) sulfate is a transition metal compound containing the metal chromium and the polyatomic ion sulfate. the oxidation state of chromium in this compound is , and the chemical formula of the compound is ( ) . reset next
Answers: 3
You know the right answer?
What should be the mole fraction of O2 in the gas mixture the diver breathes in order to have the sa...
Questions

History, 07.12.2020 23:30

Geography, 07.12.2020 23:30

Computers and Technology, 07.12.2020 23:30

Social Studies, 07.12.2020 23:30

Health, 07.12.2020 23:30

History, 07.12.2020 23:30



History, 07.12.2020 23:30


Mathematics, 07.12.2020 23:30

Mathematics, 07.12.2020 23:30

Spanish, 07.12.2020 23:30

Mathematics, 07.12.2020 23:30

Biology, 07.12.2020 23:30

Mathematics, 07.12.2020 23:30


Mathematics, 07.12.2020 23:30


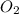 in the gas mixture the diver breathes in order to have the same partial pressure of oxygen in his lungs as he would at sea level? Note that the mole fraction of oxygen at sea level is 0.209.
in the gas mixture the diver breathes in order to have the same partial pressure of oxygen in his lungs as he would at sea level? Note that the mole fraction of oxygen at sea level is 0.209.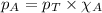 ........(1)
........(1)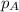 = partial pressure of oxygen at sea level = ?
= partial pressure of oxygen at sea level = ?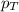 = total pressure at sea level = 1.00 atm
= total pressure at sea level = 1.00 atm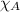 = mole fraction of oxygen at sea level = 0.209
= mole fraction of oxygen at sea level = 0.209