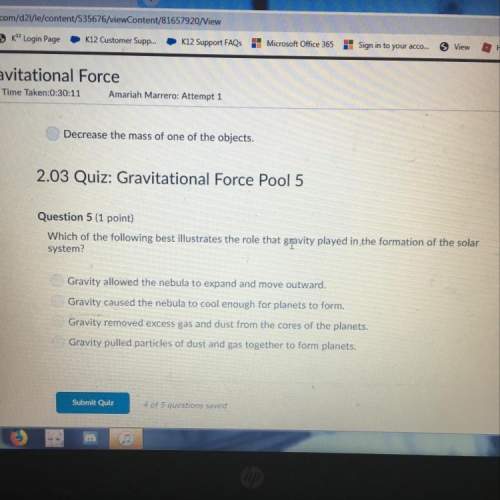
Chemistry, 12.02.2020 00:52 hadilalhjajih
Phosphine, PH₃, is a colorless, toxic gas that is used in the production of semiconductors as well as in the farming industry. When heated, phosphine decomposes into phosphorus and hydrogen gases.
4 PH₃(g) ⟶ P₄(g) + 6 H₂(g)
This decomposition is first order with respect to phosphine, and has a half‑life of 35.0 s at 953 K. Calculate the partial pressure of hydrogen gas that is present after 70.5 s if a 2.20 L vessel containing 2.29 atm of phosphine gas is heated to 953 K.

Answers: 2


Another question on Chemistry

Chemistry, 22.06.2019 04:00
Write the empirical chemical formula of calcium with a mass percent of 38.8, phosphorus with a mass percent of 20.0, and oxygen with a mass percent of 41.3.
Answers: 1

Chemistry, 22.06.2019 05:40
Fill in the coefficients that will balance the following reaction: a0cr2(so4)3 + a1agno3
Answers: 3

Chemistry, 22.06.2019 08:30
Joan writes four numbers on the board in standard form, and then she writes their scientific notation
Answers: 1

Chemistry, 22.06.2019 08:30
The characteristic of two different types of reactions are shown below. reaction a: electrons are gained by the atoms of an element. reaction b: protons are lost by the atom of and element. which statement is true about the atoms of the elements that participate in the two reactions? a: their identity changes in both reaction a and b. b: their identity changes in reaction a but not b. c: their identity changes in reaction b but not a. d: their identity remains the same.
Answers: 1
You know the right answer?
Phosphine, PH₃, is a colorless, toxic gas that is used in the production of semiconductors as well a...
Questions



Mathematics, 15.04.2021 21:20

Mathematics, 15.04.2021 21:20


Mathematics, 15.04.2021 21:20




Mathematics, 15.04.2021 21:20








Mathematics, 15.04.2021 21:20