Answers: 2


Another question on Chemistry

Chemistry, 21.06.2019 17:30
You are performing an experiment in a lab to attempt a new method of producing pure elements from compounds. the only problem is that you do not know what element will form. by your previous calculations you know that you will have 6.3 moles of product. when it is complete, you weigh it and determine you have 604.4 grams. what element have you produced?
Answers: 1

Chemistry, 21.06.2019 23:00
City a and city b had two different temperatures on a particular day. on that day, four times the temperature of city a was 8â° c more than 3 times the temperature of city b. the temperature of city a minus twice the temperature of city b was â’3â° c. what was the temperature of city a and city b on that day? city a was 5â° c, and city b was 4â° c. city a was 3â° c, and city b was â’1â° c. city a was 8â° c, and city b was â’3â° c. city a was 5â° c, and city b was â’5â° c.
Answers: 2

Chemistry, 22.06.2019 05:00
In 1901, thomas edison invented the nickel-iron battery. the following reaction takes place in the battery. fe(s) + 2 nio(oh)(s) + 2 h2o(l) fe(oh)2(s) + 2 ni(oh)2(aq) how many mole of fe(oh)2, is produced when 5.35 mol fe and 7.65 mol nio(oh) react?
Answers: 1

Chemistry, 22.06.2019 20:00
What is the molar mass of the anhydrous compound? answer using four significant figures. 36.02 g/mol 120.15 g/mol 156.12 g/mol
Answers: 1
You know the right answer?
Question 13 (3.33 points)
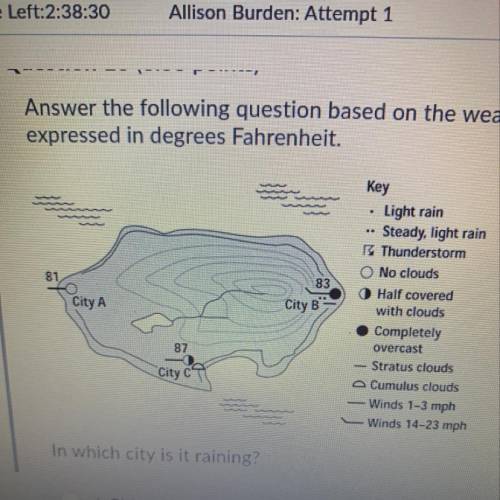
Answer the following question based on the weather chart below. Telm...
Answer the following question based on the weather chart below. Telm...
Questions

Mathematics, 27.04.2021 08:20



Chemistry, 27.04.2021 08:20

History, 27.04.2021 08:20

Health, 27.04.2021 08:20

Mathematics, 27.04.2021 08:20

History, 27.04.2021 08:20


Social Studies, 27.04.2021 08:20


Mathematics, 27.04.2021 08:20


Mathematics, 27.04.2021 08:20


Mathematics, 27.04.2021 08:20


History, 27.04.2021 08:20

English, 27.04.2021 08:20